Epistemonikos summaries
← vista completaPublished on March 15, 2017 | http://doi.org/10.5867/medwave.2017.6867
Is glucosamine effective for osteoarthritis?
¿Es efectiva la glucosamina para el tratamiento de la artrosis?
Abstract
Osteoarthritis is the most prevalent chronic articular disease, in which pain is one of the main symptoms and a major determinant of functional loss. Several therapeutic options have been proposed, including glucosamine, but its actual usefulness has not yet been established. To answer this question, we searched in Epistemonikos database, which is maintained by screening multiple databases. We identified 11 systematic reviews including 35 randomized trials answering the question of interest. We extracted data, conducted a meta-analysis and generated a summary of findings table using the GRADE approach. We concluded it is not clear whether glucosamine decreases pain or improves functionality in osteoarthritis because the certainty of the evidence is very low.
Problem
Osteoarthritis is the most common chronic joint disease in the world, and is associated with progressive and chronic joint cartilage damage. During the last few years several pharmacological treatments have emerged, including glucosamine, an endogenous aminosaccharide that would slow down the proteoglycan constituents of the articular cartilage, avoiding alterations in its structure that contribute to the degenerative process. The exogenous administration of glucosamine would allow a restoration of the cartilage, which would translate into a clinical improvement. However, its actual effectiveness in osteoarthritis has not yet been clearly established.
Methods
We used Epistemonikos database, which is maintained by screening multiple databases, to identify systematic reviews and their included primary studies. With this information, we generated a structured summary using a pre-established format, which includes key messages, a summary of the body of evidence (presented as an evidence matrix in Epistemonikos), meta-analysis of the total of studies, a summary of findings table following the GRADE approach, and a table of other considerations for decision-making.
|
Key messages
|
About the body of evidence for this question
|
What is the evidence. |
We found 11 systematic reviews [1],[2],[3],[4],[5],[6],[7], [8],[9],[10],[11], including 35 randomized controlled trials, reported in 36 references [12],[13],[14],[15],[16],[17],[18], [19],[20],[21],[22],[23],[24],[25],[26],[27],[28],[29],[30], [31],[32],[33],[34],[35],[36],[37],[38],[39],[40],[41],[42], [43],[44],[45],[46],[47]. |
|
What types of patients were included |
Twenty-seven trials included outpatients [15],[16],[17],[19], [20],[21],[22],[24],[25],[26],[27],[28],[29],[30],[31],[32], [33],[36],[37],[38],[39],[40],[41] ,[42],[43],[45],[47], four trials inpatients [12],[13],[14],[18] and in four trials this information was not described [23],[34],[44],[46]. Twenty-nine trials included patients with knee |
|
What types of interventions were included |
Twenty-nine trials used glucosamine sulfate [12],[13],[14], [15],[17],[18],[19],[20],[21],[22],[25],[26],[27],[28],[29], [31],[32],[33],[34],[36],[37],[38],[40],[41] ,[42], [43],[44], [45],[46], five trials glucosamine hydrochloride [23],[24],[30], [39],[47], and in one trial this information was not described [16]. Regarding dosing, twenty-eight trials used 1500 mg/day [14], [15],[17],[18],[19],[21],[22],[24],[25],[26],[27],[28],[29], [31],[32],[33],[34],[36],[37],[38],[39],[40],[41],[42],[43], [44],[47], two trials used 1500 mg/day for seven days followed by 400 mg/day for the rest of the treatment duration [12],[13], two trials used 2000 mg/day [30],[45], one trial 400 mg two times per week [20] and in two trials this information was not described [16],[23]. In thirty-one trials the route of administration was oral [14], [15],[17],[18],[19],[21],[22],[23],[24],[25],[26],[27],[28], [29],[30],[31],[32],[33],[34],[36],[37],[38],[39],[40],[41], [42],[43],[44],[45],[46],[47], in one trial intraarticular [16], in other trial intramuscular [20], in other trial intraarticular or intramuscular followed by oral administration [12] and in other intramuscular or intravenous followed by oral administration [13]. Four trials reported that patients received adjuvant therapy in addition to glucosamine: acetaminophen [24],[45], naproxen, ibuprofen, acetaminophen or acetyl salicylic acid [29], and exercise [44]. The duration of treatment was on average 30 weeks, with a minimum of two weeks and a maximum of three years. One trial did not report this information [23]. Twenty-nine trials compared against placebo [12],[14],[15], [16],[19],[20],[21],[23],[24],[25],[26],[27],[28],[29],[30], [31],[32],[33],[36],[38],[39],[40],[41],[42],[43],[44],[45], [46],[47], three trials compared glucosamine with ibuprofen [17],[18],[22], one trial compared to piperazine/chlorbutanol for seven days followed by two weeks of placebo [13], one trial compared glucosamine sulfate against glucosamine hydrochloride [34], and one trial against Uncaria guianens (herbal supplement known as "cat's claw") [37]. |
|
What types of outcomes |
The outcomes pooled by the systematic reviews identified were pain, Lequesne's index, WOMAC (Western Ontario and McMaster Universities Arthritis Index) pain subscale, WOMAC stiffness subscale, WOMAC functionality subscale, and WOMAC total, minimum joint space width, overall assessment of the disease by the patient and by the physician and drug toxicity. |
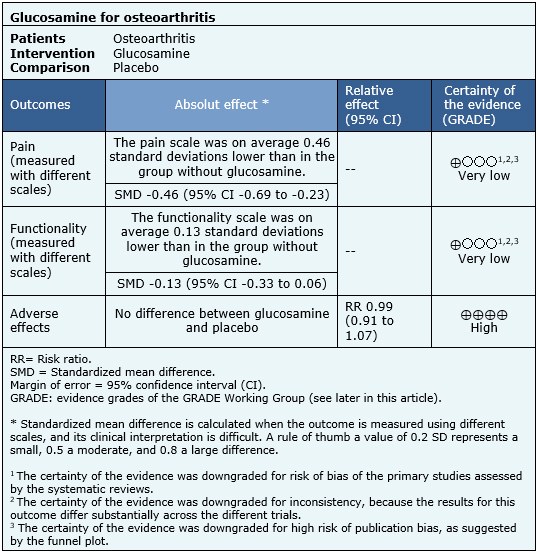
Summary of findings
The effects of glucosamine are based on 21 randomized trials that, in total, included 2,691 patients. The other trials did not report any outcome of interest, or did not present the information in a way it could be incorporated in a meta-analysis. Twenty-one trials [12],[13],[14],[15],[16],[21],[24],[25],[26],[27],[28],[29], [31],[32],[33],[36],[38],[40],[41],[44],[46] measured pain (2,691 patients) and 12 trials [24],[26],[27],[28],[29],[31],[32],[36],[38],[40],[41],[44] reported functionality (2,105 patients). Adverse effects were obtained directly from one of the systematic reviews identified [5], since it was not possible to extract more information from the other reviews. The summary of findings is as follows:
- It is unclear whether glucosamine decreases pain in osteoarthritis because the certainty of the evidence is very low.
- It is unclear whether glucosamine improves functionality in osteoarthritis because the certainty of the evidence is very low.
- Glucosamine has no adverse effects or these are minimal. The certainty of the evidence is high.

Other considerations for decision-making
|
To whom this evidence does and does not apply |
|
| About the outcomes included in this summary |
|
| Balance between benefits and risks, and certainty of the evidence |
|
| What would patients and their doctors think about this intervention |
|
| Resource considerations |
|
|
Differences between this summary and other sources |
|
| Could this evidence change in the future? |
|
How we conducted this summary
Using automated and collaborative means, we compiled all the relevant evidence for the question of interest and we present it as a matrix of evidence.
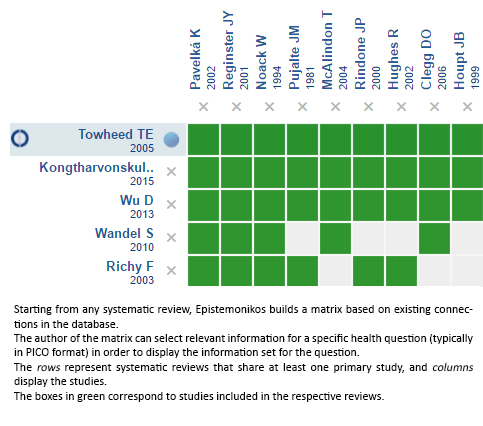
Follow the link to access the interactive version: Glucosamine for osteoarthritis
Notes
The upper portion of the matrix of evidence will display a warning of “new evidence” if new systematic reviews are published after the publication of this summary. Even though the project considers the periodical update of these summaries, users are invited to comment in Medwave or to contact the authors through email if they find new evidence and the summary should be updated earlier. After creating an account in Epistemonikos, users will be able to save the matrixes and to receive automated notifications any time new evidence potentially relevant for the question appears.
The details about the methods used to produce these summaries are described here http://dx.doi.org/10.5867/medwave.2014.06.5997.
Epistemonikos foundation is a non-for-profit organization aiming to bring information closer to health decision-makers with technology. Its main development is Epistemonikos database (www.epistemonikos.org).
These summaries follow a rigorous process of internal peer review.
Conflicts of interest
The authors do not have relevant interests to declare.

