Epistemonikos summaries
← vista completaPublished on April 5, 2017 | http://doi.org/10.5867/medwave.2017.6894
Are steroids effective in toxic epidermal necrolysis and Stevens-Johnson syndrome?
¿Son efectivos los corticoides en la necrólisis epidérmica tóxica y el síndrome de Stevens-Johnson?
Abstract
Toxic epidermal necrolysis and Stevens-Johnson syndrome are severe adverse skin reactions to medications and infections. Steroids are described as a therapeutic alternative, but their use is still controversial. To answer this question, we searched in Epistemonikos database, which is maintained by screening multiple information sources. We identified four systematic reviews including 11 primary studies answering the question of interest. We extracted data, conducted a meta-analysis and generated a summary of findings table using the GRADE approach. We concluded it is not clear whether steroids reduce mortality or hospital stay in toxic epidermal necrolysis and Stevens-Johnson syndrome because the certainty of the evidence is very low.
Problem
Toxic epidermal necrolysis and Stevens-Johnson syndrome are severe, idiosyncratic skin reactions, mainly caused by drug exposure, or secondary to bacterial and viral infections. They are both considered part of the spectrum of the same disease characterized by generalized epidermal necrosis secondary to keratinocytes apoptosis, and differentiated according to the extent of cutaneous involvement. The main pathway leading to apoptosis is the binding of FAS, a membrane receptor present in keratinocytes, with its ligand.
The therapeutic management includes general measures of support, prevention of complications and the use of different immunosuppressive drugs such as systemic steroids. However, their effectiveness in the treatment of toxic epidermal necrolysis and Stevens-Johnson syndrome is controversial and its use is associated with significant adverse effects.
Methods
We used Epistemonikos database, which is maintained by screening multiple information sources, to identify systematic reviews and their included primary studies. With this information we generated a structured summary using a pre-established format, which includes key messages, a summary of the body of evidence (presented as an evidence matrix in Epistemonikos), meta-analysis of the total of studies, a summary of findings table following the GRADE approach and a table of other considerations for decision-making.
|
Key messages
|
About the body of evidence for this question
|
What is the evidence. |
We found four systematic reviews [1],[2],[3],[4] including 11 primary studies [5],[6],[7],[8],[9],[10],[11],[12],[13], [14],[15]. None is a randomized controlled trial. |
|
What types of patients were included |
All of the studies included patients diagnosed with Stevens-Johnson syndrome (<10% of cutaneous involvement), Stevens-Johnson syndrome/toxic epidermal necrolysis overlap (10% to 30% of cutaneous involvement) and/or toxic epidermal necrolysis (>30% of cutaneous involvement). Seven studies [5],[6],[7],[8],[9],[10],[11] included patients older than 18 years, and four studies [12],[13], [14],[15] only included children. Only three studies [7],[9],[11] reported the percentage of cutaneous involvement. Six studies [6],[7],[8],[9],[10],[11] reported the etiology, with drugs being the leading cause. |
|
What types of interventions were included |
The included studies used different types of steroids, doses, route of administration, and duration. The use of oral prednisone, intravenous dexamethasone and intravenous methylprednisolone (with or without an initial bolus) was reported. Three of the studies [6],[8],[9] also incorporated intravenous immunoglobulin. |
|
What types of outcomes |
The studies measured multiple outcomes. However, those that were grouped in the identified reviews were:
|
Summary of findings
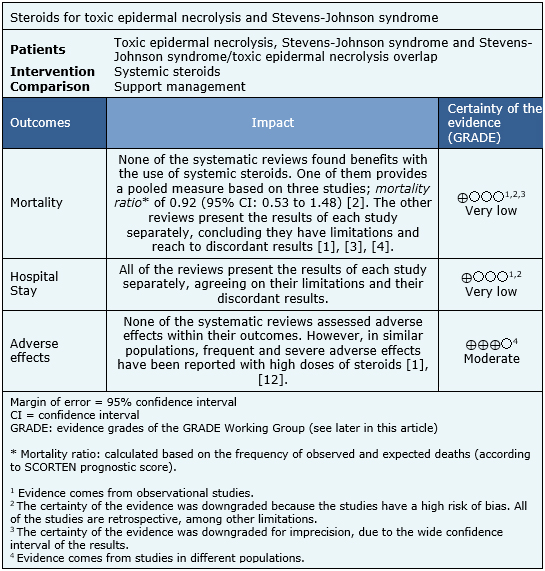
The information about the effects of steroids on toxic epidermal necrolysis and Stevens-Johnson syndrome is based on 11 observational studies [5],[6],[7],[8],[9],[10],[11],[12],[13],[14],[15]. It was not possible to extract the information of the primary studies from the systematic reviews, in order to rebuild the meta-analysis. So, the information is presented here such as presented in the systematic reviews [1],[2],[3],[4]. The summary of findings is as follows:
- It is not clear whether steroids reduce mortality in toxic epidermal necrolysis and Stevens-Johnson syndrome compared to support measures, because the certainty of the evidence is very low.
- It is not clear whether steroids reduce hospital stay in toxic epidermal necrolysis and Stevens-Johnson syndrome compared to support measures, because the certainty of the evidence is very low.
- The use of systemic steroids is associated to several adverse effects, probably frequent when treating toxic epidermal necrolysis and Stevens-Johnson syndrome. The certainty of the evidence is moderate.

Other considerations for decision-making
|
To whom this evidence does and does not apply |
|
| About the outcomes included in this summary |
|
| Balance between benefits and risks, and certainty of the evidence |
|
| What would patients and their doctors think about this intervention |
|
| Resource considerations |
|
|
Differences between this summary and other sources |
|
| Could this evidence change in the future? |
|
How we conducted this summary
Using automated and collaborative means, we compiled all the relevant evidence for the question of interest and we present it as a matrix of evidence.
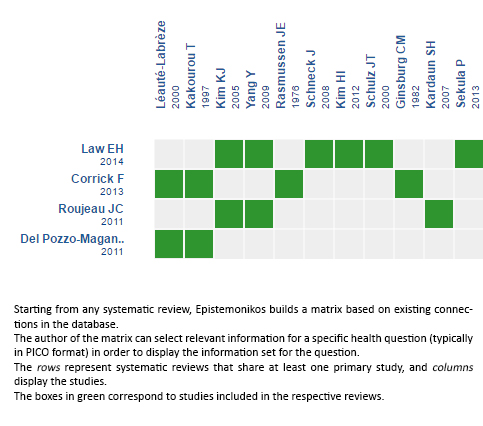
Follow the link to access the interactive version: Steroids for Stevens-Johnson syndrome and toxic epidermal necrolysis
Notes
The upper portion of the matrix of evidence will display a warning of “new evidence” if new systematic reviews are published after the publication of this summary. Even though the project considers the periodical update of these summaries, users are invited to comment in Medwave or to contact the authors through email if they find new evidence and the summary should be updated earlier. After creating an account in Epistemonikos, users will be able to save the matrixes and to receive automated notifications any time new evidence potentially relevant for the question appears.
The details about the methods used to produce these summaries are described here http://dx.doi.org/10.5867/medwave.2014.06.5997.
Epistemonikos foundation is a non-for-profit organization aiming to bring information closer to health decision-makers with technology. Its main development is Epistemonikos database (www.epistemonikos.org).
These summaries follow a rigorous process of internal peer review.
Conflicts of interest
The authors do not have relevant interests to declare.

