Epistemonikos summaries
← vista completaPublished on February 14, 2018 | http://doi.org/10.5867/medwave.2018.01.7153
Are cannabinoids effective for fibromyalgia?
¿Son los cannabinoides un tratamiento efectivo para la fibromialgia?
Abstract
INTRODUCTION Cannabinoids have been proposed as a therapeutic alternative for fibromyalgia. However, their clinical effectiveness is a matter of debate.
METHODS To answer this question we used Epistemonikos, the largest database of systematic reviews in health, which is maintained by screening multiple information sources, including MEDLINE, EMBASE, Cochrane, among others. We extracted data from the systematic reviews, reanalyzed data of primary studies and generated a summary of findings table using the GRADE approach.
RESULTS AND CONCLUSIONS We identified fifteen systematic reviews including two randomized trials overall. We concluded it is not clear whether cannabinoids have any benefit in fibromyalgia because the certainty of the evidence is very low. On the other hand, they are associated to frequent adverse effects.
Problem
Cannabinoids are a broad family of natural or synthetic compounds that can act as ligands in the cannabinoid receptors of the organism. It is postulated both CB1 and CB2 receptors have an influence on the nociceptive system [1].
The cause of fibromyalgia remains uncertain, and therapeutic response to pharmacological treatment is insufficient in a large number of patients. It is supposed cannabinoids would have a role in fibromyalgia since there is deficiency in the level of endocannabinoids in this condition [2]. It is also hypothesized that cannabinoids would reduce sensitivity of nociceptive pathways, altering cognitive and autonomous processing in states of chronic pain [3]. In addition, the distribution of cannabinoid receptors in the limbic-frontal system suggests cannabinoids may influence affective qualities of pain, which are believed to play an important role in fibromyalgia [4]. However, it is not clear what are the clinical effects of this intervention.
Methods
To answer the question, we used Epistemonikos, the largest database of systematic reviews in health, which is maintained by screening multiple information sources, including MEDLINE, EMBASE, Cochrane, among others, to identify systematic reviews and their included primary studies. We extracted data from the identified reviews and reanalyzed data from primary studies included in those reviews. With this information, we generated a structured summary denominated FRISBEE (Friendly Summary of Body of Evidence using Epistemonikos) using a pre-established format, which includes key messages, a summary of the body of evidence (presented as an evidence matrix in Epistemonikos), meta-analysis of the total of studies when it is possible, a summary of findings table following the GRADE approach and a table of other considerations for decision-making.
|
Key messages
|
About the body of evidence for this question
|
What is the evidence. |
We found 15 systematic reviews [5],[6],[7],[8],[9],[10],[11],[12],[13],[14],[15],[16],[17],[18],[19], including three primary studies answering the question of interest [20],[21],[22]. Two studies correspond to randomized trials [20],[21]. This table and the summary in general are based on the latter, since the observational study did not increase the certainty of the existing evidence or provide relevant additional information. |
|
What types of patients were included* |
Both trials included patients diagnosed with fibromyalgia according to the ACR 1990 criteria. Both trials included adults over 18 years. One trial restricted age to 70 years [20]. One trial included patients with continuous pain despite another oral medication [20] and the other trial included patients with self-reported chronic insomnia [21]. Patients with a history of substance abuse, psychotic disorders, unstable angina and previous use of cannabinoids for pain management were excluded in both trials. |
|
What types of interventions were included* |
Both trials used oral nabilone as intervention. Regarding the dose, one trial used 0,5 mg to 1 mg per day [20] and the other used 0,5 mg per day [21]. Regarding the comparison, one trial [21] compared against oral amitriptyline 10 mg daily and the other trial [20] compared against placebo. |
|
What types of outcomes |
Of the multiple outcomes measured by the randomized trials, the systematic reviews grouped them as follows:
|
* The information about primary studies is extracted from the systematic reviews identified, unless otherwise specified.
Summary of Findings
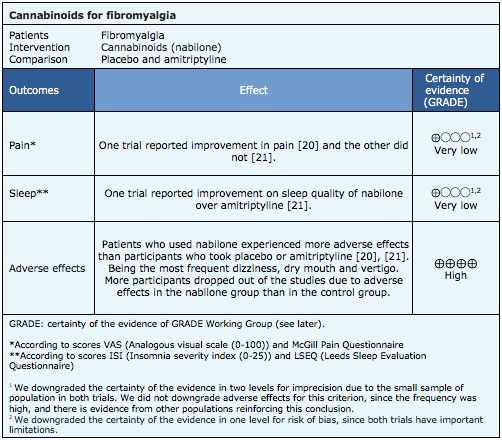
The information on the effects of cannabinoids for fibromyalgia is based on two randomized trials including 72 patients overall [20],[21]. Both trials reported the effect on pain and adverse effects. Only one trial measure sleep [21].
None of the reviews was able to extract the data in a way that could be incorporated into a meta-analysis, so the information presented below corresponds to a narrative synthesis of the information obtained from them.
The summary of findings is the following:
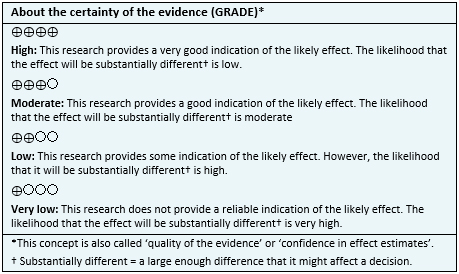
- It is not clear whether cannabinoids reduce pain, because the certainty of the evidence is very low.
- It is not clear whether cannabinoids improve sleep because the certainty of the evidence is very low.
- Cannabinoids are associated to frequent adverse effects in fibromyalgia. The certainty of the evidence is high.
Other considerations for decision-making
|
To whom this evidence does and does not apply |
|
| About the outcomes included in this summary |
|
| Balance between benefits and risks, and certainty of the evidence |
|
| Resource considerations |
|
| What would patients and their doctors think about this intervention |
|
|
Differences between this summary and other sources |
|
| Could this evidence change in the future? |
|
How we conducted this summary
Using automated and collaborative means, we compiled all the relevant evidence for the question of interest and we present it as a matrix of evidence.
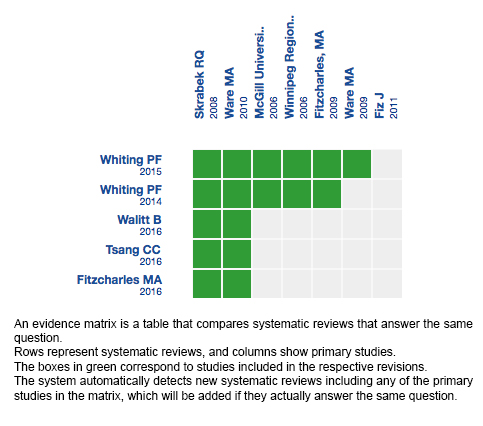
Follow the link to access the interactive version: Cannabinoids for fibromyalgia
Notes
The upper portion of the matrix of evidence will display a warning of “new evidence” if new systematic reviews are published after the publication of this summary. Even though the project considers the periodical update of these summaries, users are invited to comment in Medwave or to contact the authors through email if they find new evidence and the summary should be updated earlier.
After creating an account in Epistemonikos, users will be able to save the matrixes and to receive automated notifications any time new evidence potentially relevant for the question appears.
This article is part of the Epistemonikos Evidence Synthesis project. It is elaborated with a pre-established methodology, following rigorous methodological standards and internal peer review process. Each of these articles corresponds to a summary, denominated FRISBEE (Friendly Summary of Body of Evidence using Epistemonikos), whose main objective is to synthesize the body of evidence for a specific question, with a friendly format to clinical professionals. Its main resources are based on the evidence matrix of Epistemonikos and analysis of results using GRADE methodology. Further details of the methods for developing this FRISBEE are described here (http://dx.doi.org/10.5867/medwave.2014.06.5997)
Epistemonikos foundation is a non-for-profit organization aiming to bring information closer to health decision-makers with technology. Its main development is Epistemonikos database (www.epistemonikos.org).
Potential conflicts of interest
The authors do not have relevant interests to declare.

