Estudios originales
← vista completaPublicado el 4 de mayo de 2026 | http://doi.org/10.5867/medwave.2026.04.3151
Tendencias temporales en incidencia de cáncer en una población peruana del Seguro Social de Salud entre 2000 y 2020
Temporal trends in cancer incidence in a Peruvian population of the Social Health Insurance between 2000 and 2020
Abstract
Introduction Cancer is one of the leading causes of disease burden in Peru, with significant regional disparities in incidence and mortality. The Moquegua region has experienced a sustained increase in risk factors and cancer burden without systematic studies to characterize its epidemiological profile. The objective of this study was to estimate the temporal trend in cancer incidence and describe its characteristics in the population insured by Social Health Insurance in Moquegua between 2000 and 2020.
Methods A retrospective study was conducted based on institutional records of patients with cancer confirmed by pathological anatomy at Hospital Base II, Moquegua. Crude and age-adjusted incidence rates were calculated. Additionally, a joinpoint regression model was applied to analyze temporal trends. The analysis was stratified by sex and anatomical location. Furthermore, five-year overall survival was calculated for the most common neoplasms.
Results A total of 545 new cancer cases were identified during the study period. The overall age-adjusted incidence rate was 30.2 per 100 000 insured individuals, with a higher prevalence among women (55.2%). A significant upward shift was observed between 2013 and 2017. The most common sites were: breast (19.4%), prostate (18.0%), cervix (7.5%), colorectal (7.3%), and thyroid (7.0%). Breast cancer had the highest incidence rate; thyroid cancer had a survival rate close to 100%, while prostate cancer had the lowest overall survival rate.
Conclusions The incidence of cancer in Moquegua has shown an upward trend since 2013, possibly associated with improvements in diagnostic capacity. Rates were higher among women, with a predominance of neoplasms common at the national level. These findings underscore the need to strengthen cancer surveillance through a territorial approach and to improve access to early-diagnosis services and timely treatment.
Main messages
- This study provides new evidence on the epidemiology of cancer in a population of insured individuals in a region with unique socio-environmental conditions, revealing significant differences from the national pattern.
- The study has several limitations inherent to the use of routine healthcare registries that should be considered when interpreting the findings.
- These include the inability to generalize results, since the sample covers only the insured population; information biases due to data entry errors, inconsistencies in diagnostic coding, underreporting of cases, or other factors, and measurement bias.
Introduction
Cancer is a global public health priority due to its high burden of morbidity and mortality, as well as its economic and social impact [1]. According to GLOBOCAN 2020 estimates, cancer is the second leading cause of death worldwide, accounting for approximately 10 million deaths annually [2]. In Latin America and the Caribbean, there are nearly 1.5 million new cases and 700 000 cancer deaths each year, with incidence and mortality rates of 186.5 and 86.6 per 100 000 inhabitants, respectively. In Peru, cancer ranks among the top three causes of death, with a mortality rate of 97.9 per 100 000 inhabitants, with prostate, stomach, liver, bile duct, cervical, lung, and breast cancers being the most common [3,4]. As of 2019, it was estimated that 38.5% of cancer cases and 43.4% of cancer deaths in the country were linked to modifiable risk factors, including oncogenic infections (human papillomavirus and Helicobacter pylori), smoking, physical inactivity, harmful alcohol use, and obesity [5].
However, cancer epidemiological trends can vary significantly at the subnational level. In the Moquegua region, malignant tumors have been the leading cause of disease burden and mortality among adults since 2018, suggesting an accelerated epidemiological transition [6,7]. This phenomenon has been accompanied by an increase in the prevalence of risk behaviors such as physical inactivity, overweight, and obesity, placing Moquegua among the regions with the highest prevalence of childhood and adult obesity nationwide [8,9,10]. Additionally, sustained mining and industrial activity have contributed to environmental contamination by heavy metals, promoting the bioaccumulation of toxins in food and water, which could be associated with the higher incidence of gastric, colorectal, and breast cancers in the region [11,12,13,14]. In this context, the regional cancer pattern differs from the national profile, with a predominance of breast, cervical, skin, colorectal, and stomach cancers [10].
Peru has a fragmented healthcare system, both in terms of service delivery and health insurance. Although the Ministry of Health plays a leading role, the public sector has at least three main health insurance and service delivery systems, one of the most important being the Social Health Insurance (EsSalud), which also operates under the Ministry of Labor. The Social Health Insurance covers all dependent workers and their beneficiaries, serving approximately 30% of the Peruvian population. The Moquegua Healthcare Network of the Social Health Insurance has experienced a notable expansion in coverage, diagnostics, and access to services over the past two decades, increasing its coverage from 29.6% of the population in 2007 to 36.0% in 2017. This includes an increase in cancer care coverage. In 2020, this network provided care to more than 103 000 insured individuals, comprising a population of beneficiaries who are mostly economically active, highly educated, and exposed to multiple occupational and behavioral risk factors associated with cancer [15,16]. According to institutional reports, cancer accounted for 23% of all recorded deaths in this population [17].
Despite this significant burden, no studies have been identified that systematically and longitudinally characterize the epidemiological patterns of cancer in Moquegua. Therefore, the objective of this study was to estimate the temporal trend in incidence and describe the main epidemiological characteristics of cancer among patients treated in the Moquegua Healthcare Network of the Social Health Insurance System during the period from 2000 to 2020. This includes a description of the sociodemographic and clinical characteristics of confirmed cases, the calculation of crude and age-adjusted incidence rates by sex and anatomical location, the evaluation of temporal trends using joinpoint regression models, and the estimation of overall survival by cancer type during that period.
Methods
A retrospective observational study was conducted based on routine clinical records from the Hospital Base II Moquegua of the Social Health Insurance system. Individual data confirmed by pathology and coded according to the International Classification of Diseases, 10th Revision (ICD-10) were used. This enabled the construction of a consistent time series and the evaluation of annual variation patterns in relation to demographic and clinical characteristics. The methodological approach made it possible to examine the evolution of the oncological burden in a real-world healthcare setting, considering the specific features of institutional recording systems [18].
The study was carried out at Hospital Base II, Moquegua, the main oncology referral center of the Social Health Insurance system in the studied region. It is located in the district of Moquegua, province of Mariscal Nieto, in southern Peru. The estimated population in this region, according to the 2017 National Census, was 174 863 inhabitants, with a distribution of 50.4% men and 49.6% women, and an age structure composed of 23.3% under 15 years, 67.4% between 15 and 64 years, and 9.3% older adults [19].
All records of patients with a confirmed diagnosis of cancer treated in the Moquegua Healthcare Network between January 1st, 2000, and December 30th, 2020, were included; these were reviewed in October 2022. For the period 2000 to 2019, records from the Hospital Management System were used, while for 2020, records from the Intelligent Health Services Management System were employed. This platform progressively replaced the Hospital Management System as the institutional clinical recording procedure. Both systems constitute the only official sources of oncological data within the healthcare network; therefore, no additional databases were used. Only cases with a definitive diagnosis confirmed by pathology and coded according to ICD-10, within the range C00 to C96, were included. Records with incomplete data, duplicates, presumptive diagnoses, and cases permanently referred to other regions were excluded.
Sociodemographic (age, sex) and clinical variables (type and anatomical location of cancer, year of diagnosis, and year of death) were collected. The measurement of these variables provided information for case characterization; the year of diagnosis was used to measure incidence, and together with the year of death, to calculate five-year overall survival. The information was systematized in a structured database.
Statistical analysis
Crude and age-adjusted incidence rates were calculated, using the World Health Organization (WHO) standard population as reference [20]. Age-adjusted incidence rates were calculated overall, by sex, and according to the five most frequently diagnosed neoplasms. Likewise, five-year overall survival rates were calculated for these neoplasms, considering the individual dates of diagnosis and death.
Fourteen age groups were established in five-year intervals (0 to 4; 5 to 9; 10 to 14; 15 to 19; 20 to 24; 25 to 29; 30 to 34; 35 to 39; 40 to 44; 45 to 49; 50 to 54; 55 to 59; 60 to 64; 65 and over). Incidence rates were also standardized using the direct method, with the WHO standard population as reference [20].
Subsequently, a joinpoint regression analysis was performed, with stratification by sex, to estimate time intervals and annual percent change (APC) in the calculated rates, the average annual percent change over the entire study period, and their corresponding 95% confidence intervals. Additionally, the average annual percent change (AAPC) was calculated as an indicator of the mean annual change over the study period. The AAPC is defined as the geometric mean of the APCs, weighted by the length of each segment (separated by joinpoints) [21].
For model fitting, according to the guidelines of the Division of Cancer Control & Population Sciences of the National Cancer Institute (United States), the weighted Bayesian Information Criterion (BIC) was used. This allows selection of the best model and the appropriate number of joinpoints by penalizing model complexity (i.e., more parameters imply greater complexity) to avoid overfitting. No evidence of autocorrelation was found, allowing the use of a non-correlated error model. A p value < 0.05 was considered statistically significant.
Five-year overall survival was estimated using the Kaplan–Meier method, defining death from any cause as the event. Follow-up time was calculated at the individual level as the difference between the date of confirmed diagnosis and the date of death. In the absence of the event, cases were administratively censored at the study closing date (October 2022) [22]. Since the data source recorded follow-up time in calendar years, particularly in older records, a continuity correction was applied to handle tied times and short-duration events, assigning 0.5 years to cases in which death occurred in the same year as diagnosis, in accordance with methodological recommendations for survival analysis in cancer registries with incomplete temporal information [23]. Cumulative probabilities of five-year overall survival and their corresponding 95% confidence intervals were estimated and reported, along with survival functions using Kaplan–Meier curves.
These analyses were conducted using R 4.5.1 ® in RStudio 2025.09.1 ® (packages: tidyverse and survival), and for joinpoint regression, the Joinpoint Regression Program, Version 5.2.0.0, Statistical Research and Applications Branch, National Cancer Institute ®.
Ethical considerations
The study protocol was approved by the Institutional Research Ethics Committee of Hospital Base II, Moquegua (Letter No. 685-DRAMOQ-ESSALUD-2024). As anonymized clinical records were used, informed consent was waived. Data confidentiality was ensured in accordance with current ethical and legal standards for health research.
Results
Between January 2000 and December 2020, 545 new cases of cancer were confirmed among patients treated in the Moquegua Healthcare Network, of which 301 (55.2%) were women and 231 (44.8%) were men. Of the total records initially identified, 20 were excluded due to incomplete or duplicated information or failure to meet established diagnostic criteria, thus forming the final dataset used for the analyses. The age-adjusted incidence rate for the entire period was 30.2 cases per 100 000 inhabitants per year. The time series showed an initial incidence of 11.6 cases per 100 000 inhabitants in 2000, increasing to 59.7 cases per 100 000 inhabitants in 2020.
The joinpoint regression analysis identified two breakpoints in 2013 and 2017 (average annual rate of change: 8.55; 95% confidence interval: 3.88 to 15.82; p = 0.001). This allowed the study period to be divided into three stages: from 2000 to 2013, a non-significant downward trend was observed (annual percentage change: -0.59; 95% confidence interval: -15.52 to 11.16; p = 0.940); while between 2013 and 2017, a significant increase in incidence was observed (annual percentage change: 73.87; 95% confidence interval: 40.32 to 137.49; p = 0.001); followed by a period of decreasing incidence from 2017 to 2020 (annual percentage change: -15.23; 95% confidence interval: -38.58 to 2.52; p = 0.086) (Figure 1).
Joinpoint regression of age-adjusted cancer incidence rates.
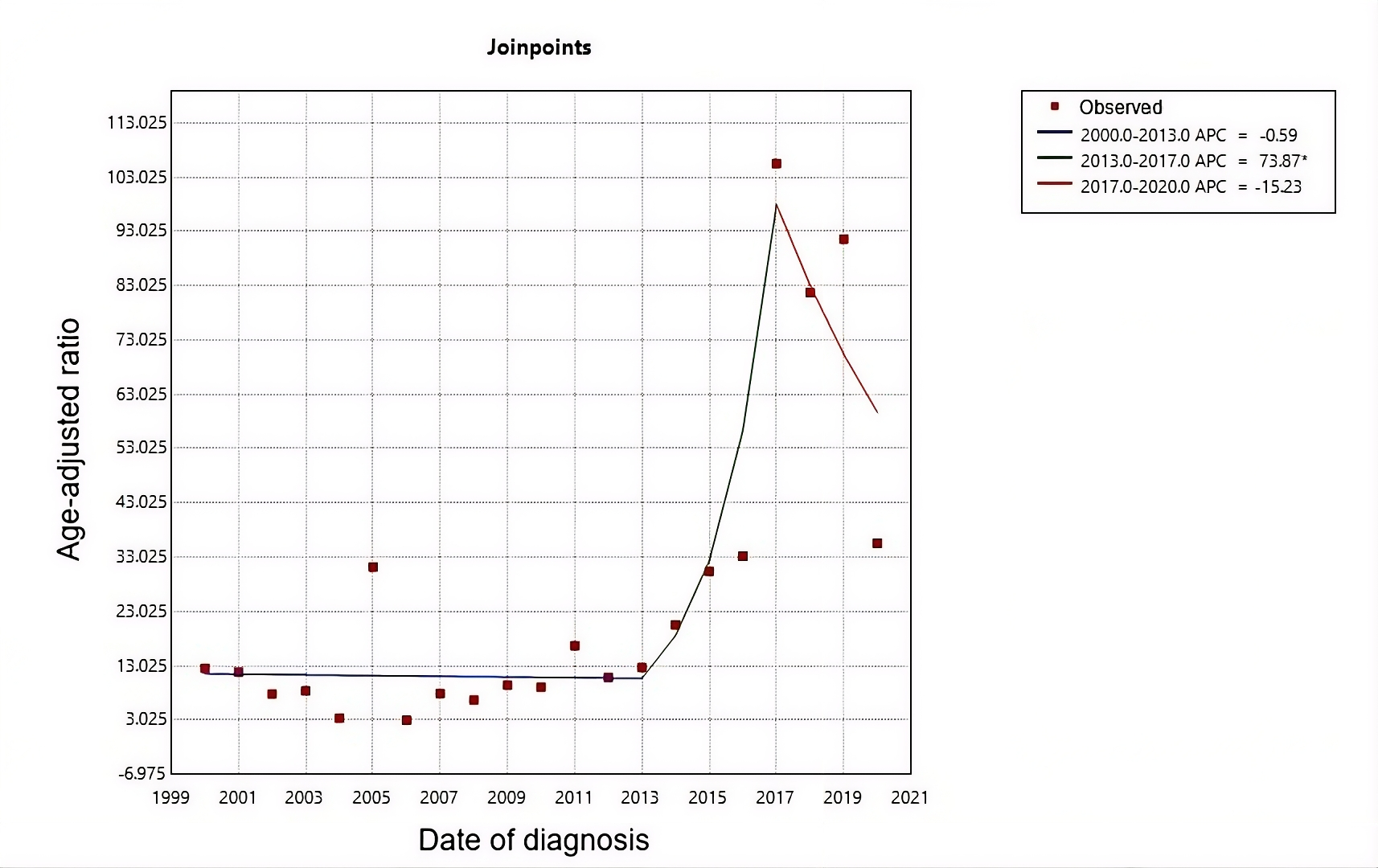
(*) Indicates that the annual percentage change is significantly different from zero at a significance level of α = 0.05.
Source: Author’s own analysis using Joinpoint Regression Program 5.4.0®.
In the analysis by gender, the age-adjusted incidence rate was 6.58 cases per 100 000 inhabitants per year for women and 5.32 cases per 100 000 inhabitants per year for men. In women, the joinpoint regression showed a change in 2011: from 2000 to 2011, there was a non-significant downward trend (annual percentage change: -12.79; 95% confidence interval: -69.19 to 5.03; p = 0.162), while between 2011 and 2020 a significant increase was observed (annual percentage change: 22.16; 95% confidence interval: 9.08 to 100.10; p = 0.015). The average annual percentage change for women over the entire period was 1.49% (95% confidence interval: -4.89 to 12.01; p = 0.390). For men, the model did not identify significant breakpoints, and the estimated average annual percentage change was 4.27% (95% confidence interval: -3.82 to 23.38; p = 0.188) (Figure 2).
Joinpoint regression of age-adjusted cancer incidence rates by gender.
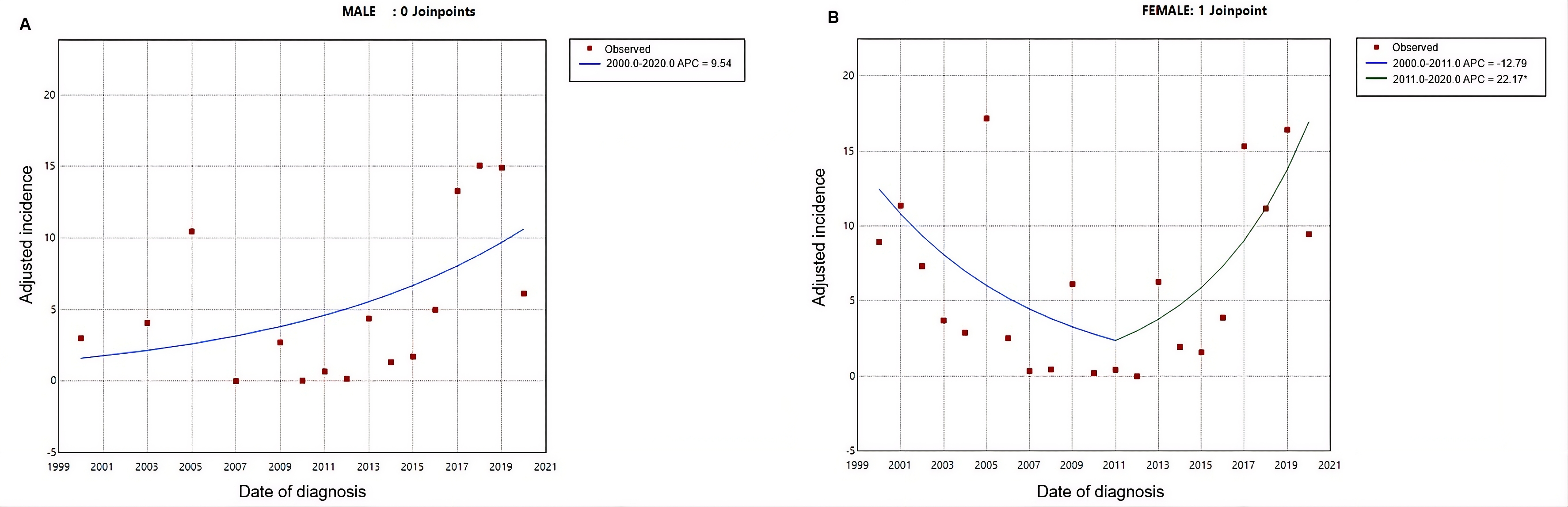
Data obtained from patients treated in the Moquegua Healthcare Network (2000–2020).
(*) Indicates that the annual percentage change is significantly different from zero at the 0.05 significance level.
Source: Author’s own analysis using Joinpoint Regression Program 5.4.0®.
Thirty anatomical sites of malignant neoplasms were identified, with the most common being: breast (19.4%), prostate (18.0%), cervix (7.5%), colorectal (7.3%), and thyroid (7.0%).
In terms of age-adjusted incidence over the entire study period, breast cancer had the highest rate at 13.15 cases per 100 000 inhabitants per year, followed by prostate cancer (10.98 per 100 000 inhabitants per year). Five-year overall survival by cancer type ranged from 97.4% (thyroid) to 74.7% (colorectal), as shown in Table 1.
The five-year overall survival in the study population was 74.8% (71.0% to 78.7%). The overall survival curves and those by cancer type are shown in Figure 3.
Overall survival curves for the five most common types of cancer.
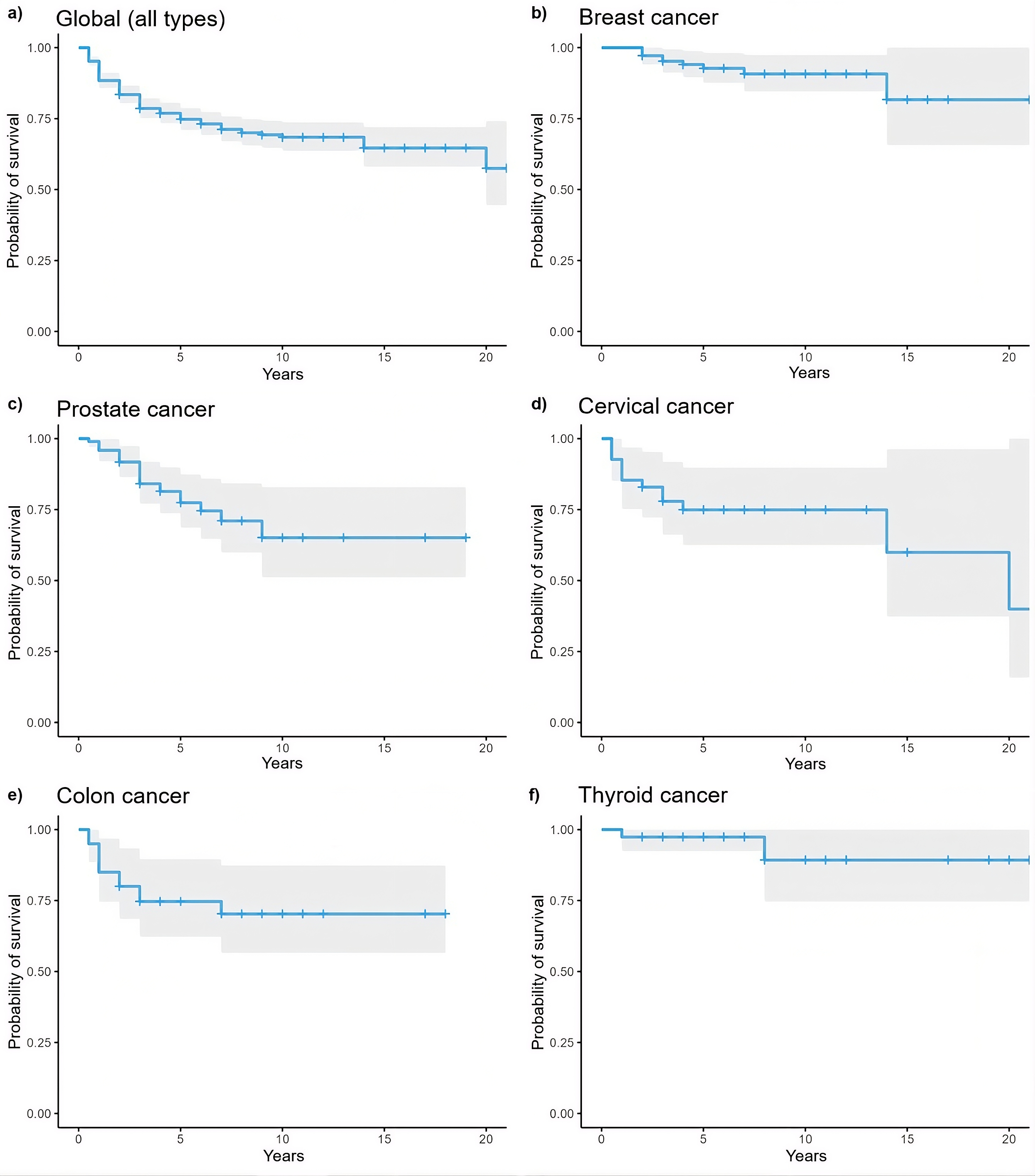
Source: Prepared by the authors.
Discussion
This study estimated an age-adjusted incidence rate of 30.2 cases per 100 000 beneficiaries of the Moquegua Healthcare Network of the Social Health Insurance System between 2000 and 2020. Furthermore, rates higher than those reported by the National Center for Epidemiology, Prevention, and Disease Control for the Moquegua region in 2020 were observed (16.2 per 100 000 inhabitants versus 59.7 per 100 000 inhabitants in this study) and 24.4 per 100 000 inhabitants for the year 2024 [4]. Although the temporal trend observed in this study suggests a decline in incidence since 2018, which would explain the results obtained by the National Center for Epidemiology, Prevention, and Disease Control, it must be noted that these discrepancies may be associated with differences in the population source (Social Health Insurance enrollees versus the general population) and potential underreporting in the early years of the analysis period.
In this context, any differences between the two populations should be interpreted with the understanding that the population covered by Social Security Health Insurance tends to have a higher proportion of adults and older adults, exhibits greater continuity of care, and has more systematic access to specialized services and confirmatory diagnostic procedures compared to the general population of the region [24]. These structural characteristics typically favor greater capture of incident cases and more timely detection of neoplasms, particularly in health systems with insured coverage [24]. Furthermore, the general population includes groups with greater socioeconomic heterogeneity and persistent barriers to geographic, economic, or administrative access to health services. This can result in underdiagnosis or delays in case confirmation [25]. Although these differences—related to demographics, socioeconomic status, and access to services—are plausible from a theoretical standpoint and have been described in the literature, they were not empirically evaluated in the present analysis.
The join-point regression analysis identified a turning point in 2013, after which a significant increase in cancer incidence was observed. This trend is consistent with the findings of Soto-Becerra et al. (2023), who reported a sustained increase in cancer prevalence among Social Health Insurance enrollees between 2020 and 2022 [26]. In contrast, the downward trend observed between 2000 and 2013 differs from reports by the Ministry of Health, which indicated a national increase in new cases during that period [27], although it should be noted that this difference was not statistically significant in this study. This pattern suggests that the initial phase of the analysis may have been influenced by limited diagnostic capacity or deficiencies in registration and reporting. It is important to note that, starting in 2011, Peru implemented the Budgetary Program for Cancer Prevention and Control, which led to improvements in infrastructure, human resources, and the acquisition of diagnostic technology [28]. In the Moquegua region, these national policies led to the acquisition of equipment such as mammography machines and the establishment of specialized services. This may have contributed to the observed increase in subsequent incidence rates.
During the study period, a higher proportion of cases was observed among women (55.2%), with an incidence rate higher than that of men (6.58 versus 5.32 per 100 000 inhabitants, respectively). This pattern can be explained in part by the high prevalence of cancers specific to women, such as breast and cervical cancer [29]. However, the persistence of barriers to access to prevention and timely diagnostic services among Peruvian women must also be considered, especially in rural areas and among those in socially vulnerable conditions [30]. Although no Peruvian studies were identified that directly compare the temporal trends in cancer incidence by sex, international research such as that by Kehm et al. (2019) has documented a sustained upward trend in women, while rates in men have shown greater stability over time [31]. These findings support the hypothesis that social and structural determinants, rather than biological factors, may be influencing the observed gender-based variations.
The most common anatomical sites in this study were the breast, prostate, cervix, colorectal tract, and thyroid. These findings partially coincide with national reports from the Ministry of Health, which rank breast, cervical, stomach, and colorectal cancers among the most common [32]. Although there is year-to-year variability, available data from previous years have also shown a high incidence of thyroid cancer [10]. This pattern has been observed in other regions of Latin America, where the disease burden of breast and prostate cancer continues to rise in a manner similar to this study. Similarly, a regional analysis based on data from 2024 identified cervical cancer as the most common, followed by breast, skin, colorectal, and stomach cancers [33]. These findings suggest that although the order of frequency may vary slightly from year to year, the anatomical sites tend to remain consistent over time in cancer surveillance systems, except for thyroid cancer. In this regard, it is worth noting that a previous study reported a significant increase in the national incidence of this cancer, rising from 4.7 to 15.2 cases per 100 000 inhabitants between 2005 and 2016 [34]. This increase has been attributed, in large part, to improvements in early diagnostic capabilities, particularly the widespread use of thyroid ultrasound, as well as to its high five-year survival rate (97.4%) [35].
The analysis of overall survival, as shown by Kaplan–Meier curves (Figure 3), revealed clear differences by anatomical location. Consistent with the literature, thyroid cancer exhibited a five-year overall survival close to 100%, with a nearly flat curve throughout follow-up, reflecting its generally indolent nature and the impact of early detection. Similarly, breast cancer showed a high probability of survival, with declines occurring later over time, whereas prostate and colorectal cancers showed more pronounced decreases during the first years after diagnosis, as reported in other middle-income countries [36,37]. Prostate cancer was the anatomical site with the highest incidence rate. Although an increase in the detection of new cases was observed, studies such as that by Torres-Román et al. (2022) report a significant decrease in prostate cancer mortality in Moquegua between 2005 and 2017 (annual percent change: −6.0; 95% confidence interval: −11.4 to −0.2; p < 0.05) [38]. The findings reported in that study may also be explained by increased early detection due to improved diagnostic capacity in the regional health system, particularly within the Social Health Insurance system, as well as by the implementation of more effective therapeutic protocols since 2015 [39,40].
The present study has several limitations inherent to the use of routine healthcare records that should be considered when interpreting the findings. First, the information comes exclusively from administrative systems of the Social Health Insurance system, which means the cases correspond only to the insured population and do not represent the entire regional population, limiting the generalizability of the results to this specific group. In addition, the quality and completeness of records may vary over the analyzed period, particularly in the earlier years, potentially introducing information bias due to data entry errors, inconsistencies in diagnostic coding, or underreporting of cases. All of these factors may have influenced the observed trends. Similarly, variations in diagnostic capacity, availability of oncology services, and the transition between record systems (Hospital Management System and Intelligent Health Services Management System) may have affected the timeliness and accuracy of new case reporting. Finally, although five-year overall survival was estimated from available institutional records, data on the underlying cause of death and systematic follow-up were unavailable to assess it precisely, which should be considered a source of measurement bias. While an administrative censoring approach was assumed in these calculations, the results should be interpreted as preliminary, given the lack of information on other types of censoring (for example, cases referred to other healthcare facilities for definitive treatment).
Despite these limitations, the study provides a robust longitudinal overview of oncological patterns in an insured population over a 20-year period. Consequently, it offers useful evidence to strengthen epidemiological surveillance and the planning of oncology services in the region.
Furthermore, this study provides valuable data on age-adjusted incidence rates, allowing valid comparisons with other sources of information at local, regional, and national levels. This type of evidence is essential for strengthening cancer epidemiological surveillance systems in the country. Future research is recommended to further analyze oncological burden using a territorial approach, as regional patterns (such as those observed in Moquegua) do not always reflect national trends. In this regard, the use of standardized methodologies, such as calculating adjusted rates and applying joinpoint regression models, may help generate more accurate projections and more useful inputs for public health decision-making.
Conclusions
This study found a cumulative incidence of 10.6 cancer cases per 100 000 beneficiaries of the Moquegua Healthcare Network of the Social Health Insurance System during the period from 2000 to 2020. Starting in 2011, a turning point in the temporal trend was identified, marking the beginning of a sustained increase in incidence rates. This trend could be related to improvements in diagnostic capacity and access to specialized oncology services. Rates were consistently higher in women, which corresponds to the high frequency of female-specific neoplasms. The most common anatomical sites included the breast, cervix, prostate, colorectal tract, and thyroid. Furthermore, analysis of five-year overall survival showed significant differences by anatomical site, with higher survival probabilities for thyroid and breast cancer, and intermediate values for prostate and colorectal cancers. This reflects the clinical and prognostic heterogeneity of the neoplasms studied. These findings reveal a distinct regional oncological epidemiological profile, highlighting the need to strengthen cancer surveillance with a subnational focus. Similarly, it is imperative to design specific interventions based on the demographic and environmental characteristics of each region.

