Estudios originales
← vista completaPublicado el 27 de febrero de 2026 | http://doi.org/10.5867/medwave.2026.01.3164
Programación remota de implantes cocleares en Chile logra resultados comparables a la atención clínica presencial
Telehealth cochlear implant programming in Chile yields outcomes comparable to in-person clinical care
Abstract
Introduction Remote programming can improve access and help overcome barriers related to distance, cost, and travel in the follow-up care of cochlear implant users. This study evaluates the ease of use, effectiveness, and acceptability of the remote programming system for cochlear implant users.
Methods Cross-sectional study with 14 adult participants. The first phase included in-person and remote programming sessions at the hospital, while the second focused on remote programming sessions in participants' homes. In each phase, speech perception with the created programs was evaluated in a soundproof booth. Subjective feedback was obtained from audiologists and users through customized questionnaires.
Results Speech scores indicated that both maps created remotely at the hospital (first phase of the study) and those created at home (second phase of the study) provided comprehension of phrases and words that was not inferior to that of the map created in person. Scores for disyllabic words with audiovisual cues, obtained through remote programming at home, were not inferior to those obtained through remote programming at the hospital. In the subjective evaluation, both participants and audiologists indicated that the experience with remote programming was satisfactory.
Conclusions Remote programming was found to be as effective as in-person programming, as it provided access to the programming parameters and objective measures used in in-person sessions. Speech recognition results were similar for programs created in person and remotely. Participants indicated that the remote programming method and the resulting program were useful and acceptable, and that they experienced similar ease and quality of communication.
Main messages
- Remote programming has the potential to make cochlear implant follow-up more time- and cost-effective.
- Remote programming of the cochlear implant was as effective as in-person programming.
- Maps created remotely provided speech understanding that was no worse than that of maps created in person.
- Further research should explore the relationship between speech recognition outcomes and the need for in-person support during remote cochlear implant programming, as well as assess long-term outcomes and sustained adoption of this model of care.
Introduction
Advances in cochlear implant technologies, together with the expansion and optimization of surgical and audiological practices over the last few decades, have led to an increasing number of people pursuing cochlear implants as a treatment for severe to profound hearing loss. After implant surgery, access to periodic clinical follow-up is essential for providing optimal device programming and appropriate therapeutic care [1]. Despite this, studies have shown that recipients' adherence to postoperative follow-up is generally low and declines significantly with increasing time since activation [2].
To optimize the benefits of cochlear implant intervention and leverage resources effectively, it is essential to understand the barriers patients face in attending follow-up appointments. Geographical and socioeconomic barriers have been identified as impediments to accessing specialized centers [3]. In addition, with increasing life expectancy and changes in candidacy criteria, the number of cochlear implant surgeries performed in older adults, has risen significantly [4]. Thus, an increasing number of elderly people are being treated in health services, who often have difficulty getting around and require family members or caregivers to accompany them to the CI center.
Telehealth has emerged as a technological evolution in healthcare, aiming to expand services and provide improved care at reduced costs [5]. The demand for teleconsultation has developed following the COVID-19 pandemic [6]. In audiology, virtual services are available in various areas, including hearing aid and cochlear implant fitting [5,7,8]. Remote programming of cochlear implant sound processors has shown to be feasible and to be as safe and effective as in-person programming [3,9,10,11,12,13]. It therefore has the potential to expand access to follow-up care and reduce cost and travel time for patients and their families or caregivers, especially those living far from specialized centers or with reduced mobility [9,10,11,13,14]. In addition, patients have reported being satisfied with the care provided remotely [11,14].
The first telehealth methodology for remotely programming cochlear implants in clinical practice received approval from the U.S. Food and Drug Administration in 2017 [15]. This system allowed for synchronous remote programming by clinicians via mailing a tablet loaded with clinical fitting software and video conferencing software and hardware, a programming interface, and a programming cable to the remote site where the recipient was located.
In 2023, a novel, smartphone-based remote programming system was approved by the FDA [16]. This system comprises a dedicated app (AB remote support) for Bluetooth-enabled iOS and Android smartphones that enables cochlear implant recipients (ages 13 years and older) to participate in remote cochlear implant programming from anywhere with a stable internet signal (Wi-Fi, LAN, 4G or greater; at least five megabytes per second uploads and downloads speeds). This app is compatible with sound processors and hearing aids’ universal Bluetooth connectivity that allows wireless two-way streaming from any Bluetooth device. Hence, the recipient’s smartphone can function like a programming interface, thereby eliminating the need for additional hardware for remote programming. On the audiologist’s side, the system comprises a clinician-facing programming software that enables synchronous remote programming and real-time audio and video communication with the patient without additional software. Hearing health professionals can remotely adjust the cochlear implant programming parameters, and measure impedance and compound evoked action potential measurements just like they can when the patient is in front of them in the clinic.
This study aimed to evaluate the usability, effectiveness, and acceptability of the novel smartphone app-based remote programming system from the perspective of recipients and their audiologists in Chile.
Methods
Study design
This cross-sectional, prospective, within-subjects study was approved by the Ethics Scientific Committee of Hospital Clínico de la Universidad de Chile under the number. 73 – Oct 19th, 2023, and was conducted following the Declaration of Helsinki. All participants signed an informed consent form before initiation of the study procedures.
Inclusion criteria were as follows: (1) recipients of Advanced Bionics (Valencia, CA, USA) cochlear implants (CII or later), (2) over 13 years old, (3) history of post-lingual onset of sensorineural hearing loss, (4) at least six months of cochlear implant use experience, and (5) native Spanish speakers. Participants with neurological disorders and a history of facial nerve stimulation or other non-auditory percepts were excluded from the study.
Participants
Cochlear implant recipients were invited to participate by audiologists during their scheduled clinical appointments at an academic implant program in Santiago, Chile. Fourteen adult unilateral cochlear implant recipients met the inclusion criteria and agreed to participate in the study (Table 1). All participants had a history of regular follow-up appointments and at least one month of experience with the Advanced Bionics Marvel sound processor. All participants were implanted with a Hi-Res Ultra implant and a Slim J electrode array. While seven out of the 14 study participants were bimodal, the study focused on programming cochlear implant sound processors only.
Study devices
The study comprised two phases: phase 1 (in-person and remote-at-hospital programming) and phase 2 (remote at-home programming). Study-dedicated Marvel CI M90 sound processors were used for both phases. During phase 1, two processors were used with each participant, one for in-person programming and the other for the remote at-hospital programming. The smartphone used for remote programming at the hospital was a study-specific iPhone 13. For remote at-home programming, participants used their smartphones (iOS and Android).
All implant programming (in-person and remote) was conducted by an audiologist using the Target cochlear implant fitting software (version 1.5). During the in-person programming session, a NoahLink interface was used to wirelessly connect with the implant processors. Recipients used the smartphone-based AB remote support app to participate in both remote programming sessions.
In addition, the National Institute for Occupational Safety and Health Sound Level Meter (NIOSH SLM) for iOS and the Decibelios for Android app were used to measure ambient noise levels at the location where the implant recipient was present during all programming sessions [17]. Finally, the ‘SpeedTest by Ookla’ app [18] was used to measure the upload and download internet speeds via the smartphone used for remote programming at the two remote locations.
Procedures
The overview of study procedures is shown in Figure 1.
Study procedures.
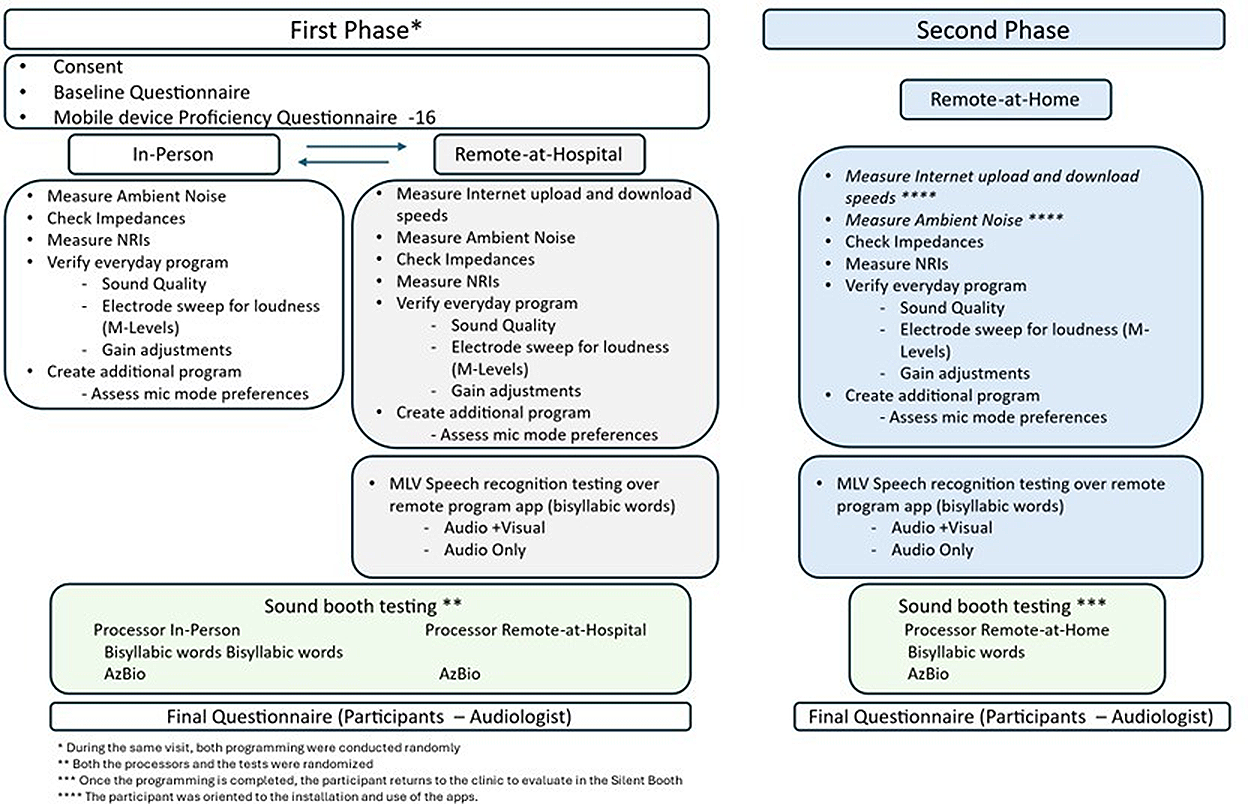
Source: Prepared by the authors based on the protocol of the study.
The same programming protocol was used for all sessions duringboth phases, as indicated in Figure 1.
During the first phase of the study, programming was conducted in two different rooms in the hospital. The in-person programming was conducted in a room typically used for this purpose in the clinic. The remote-at-hospital programming was conducted with the participant in a larger conference room with stable Wi-Fi, while the programming audiologist remained in the clinical room. As this was their first experience with remote programming, participants were provided with instructions for using the remote support app, and a second audiologist accompanied them in case assistance was needed. The order of the in-person and remote programming in this phase was randomized across the participants.
In the second phase of the study, 10 of the 14 participants underwent remote programming at their home (‘remote-at-home’), while the audiologist was in the hospital’s cochlear implant programming room. The withdrawal was due to one participant experiencing an Internet connection failure during the remote session at home, and the other three were not available for the remote at-home programming session on the scheduled date.
In addition to the remote support app, NIOSH SLM and SpeedTest by Ookla apps [18] were installed on the participants’ cellphones, and instructions for their use were provided. This enabled participants to measure ambient noise levels and internet speeds before the remote programming session began at home.
A baseline questionnaire was used to gather study-relevant demographic data, hearing history, and information about the burden and cost of travel to the clinic. Participants also answered questions regarding the cost and burden of attending follow-up visits in person (distance, time, cost, time off from work or school), access to resources needed for smartphone app-based remote programming, and their views on the use of remote programming. The questionnaire also asked participants to rate their ability to understand speech in quiet and in noise on a five-point scale (1 = very poor, 5 = very good). Finally, the participants completed a perceived benefits questionnaire, rating their agreement with statements about their remote programming experience on a five-point scale (1 = strongly disagree, 5 = strongly agree), and providing additional input at the end of both phases of the study.
Cochlear implant clinician questionnaires: The two audiologists who participated in the study completed a per-subject questionnaire to give feedback on their experience with remote programming sessions. They answered questions regarding communication with the CI recipient, satisfaction with the program created, overall session, and ease of remote programming use. Technical challenges experienced during the remote session were also asked in the study. The audiologists also completed an end-of-study questionnaire in which they rated statements about their remote programming experience on a five-point scale (1 = strongly disagree, 5 = strongly agree) and provided additional input on the applicability of remote programming to their clinical practice.
Speech perception: Speech perception was evaluated in each remote programming session with bisyllabic words presented at 65 dB via the AB support remote app, using monitored live voice. Two lists of 25 words each were presented: one with audio-only (camera switched off during remote communication) and the other with audiovisual cues (camera switched on), while voice level was monitored using the NIOSH SLM app. After each list was administered, the percentage of words correctly repeated by the participant was calculated.
Speech perception was also evaluated in quiet in a sound booth at the end of each phase. In phase one, testing was done for the in-person and remote at-hospital programs in a randomized order. In phase two, testing was done with the remote at-home program. Testing included a monitored live voice, an audio-only presentation of 25 bisyllabic words, and one list of recorded Spanish AzBio sentences [20,21] at 65 dBA from a speaker positioned at 0° azimuth. After the administration of each list, the percentage of words or sentences correctly repeated by the participant was calculated.
Statistical analysis
Qualitative data were summarized as frequencies and percentages. Quantitative outcomes were analyzed using nonparametric methods due to the small sample size.
Environmental noise levels across study phases were compared using the Friedman test. In contrast, the internet connection speed between the two conditions (remote-at-hospital and remote-at-home) was analyzed using the Wilcoxon signed-rank test. Speech perception outcomes, including AzBio sentence recognition and monitored live voice bisyllabic word scores, were analyzed using the Wilcoxon signed-rank test for two conditions for phase 1 comparisons (in-person and remote at-hospital) and the Friedman test for comparisons across three conditions for phase 1 and 2 comparisons (in-person, remote at-hospital, and remote at-home), followed by Conover’s post hoc test when appropriate. Subjective feedback from cochlear implant recipients was also analyzed using the Wilcoxon signed-rank test.
For all inferential analyses, appropriate nonparametric effect size measures were reported (rank-biserial correlation (rrb) for Wilcoxon tests and Kendall’s W (W) for Friedman tests). For all analyses, p < 0.05 was considered significant.
Results
Ambient noise levels and internet speeds
Table 2 presents ambient noise levels measured during each programming session (in-person, remote at-hospital and remote at-home) and internet speeds during the two remote programming sessions. No significant differences were observed between sessions (p > 0.05).
Proficiency with smartphone-related tasks
Average global Mobile Device Proficiency Questionnaire-16 (MDPQ-16) ratings for proficiency across the eight categories of smartphone tasks ranged between 1.85 and 5 (mean = 3.8; SD = 1.16), with one participant (7.1%) rating less than two (i.e., “not at all” proficient), five participants (35.7%) between two and three (i.e., between “not at all” and “not very easily” proficiency levels) and eight participants (57.1%) at more than an a global rating of four (i.e., “somewhat easily” to “very easily”).
Speech perception
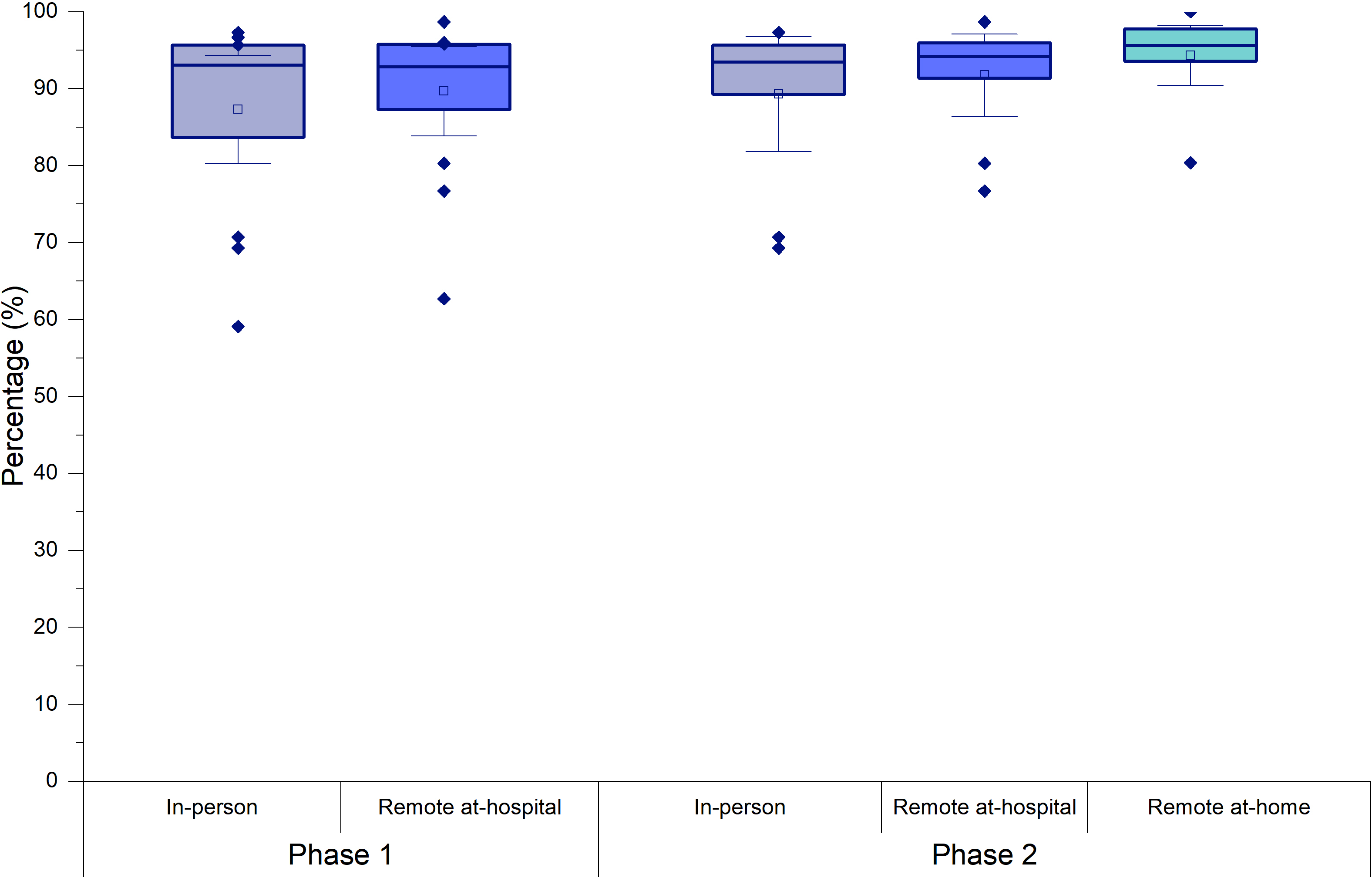
Speech recognition obtained with AzBio sentences in phase 1 were statistically better (n = 14; z = -2.103; p = 0.035* rrb = -0.638) with the remote at-hospital program (mean = 89.67%; SD = 10.07; 95% CI = 83.85 to 95.48) than the in-person program (mean = 87.31%; SD = 12.15 95% CI = 80.29 to 94.32). For the ten participants who took part in both phases, statistically better results (n = 10; t = 2.449; p = 0.025*; rrb = -0.745) were observed with the remote at-home program (mean = 94.30%; SD = 5.44; 95% CI = 90.41 to 98.19) than in the in-person program (mean = 89.30%; SD = 10.42; 95% CI = 81.85 to 96.76). The scores with the remote at-hospital program were not significantly different than those with the in-person and remote at-home programs (p > 0.05) (Figure 2).
Percentage of correct sentences on the AzBio test in phases 1 and 2.
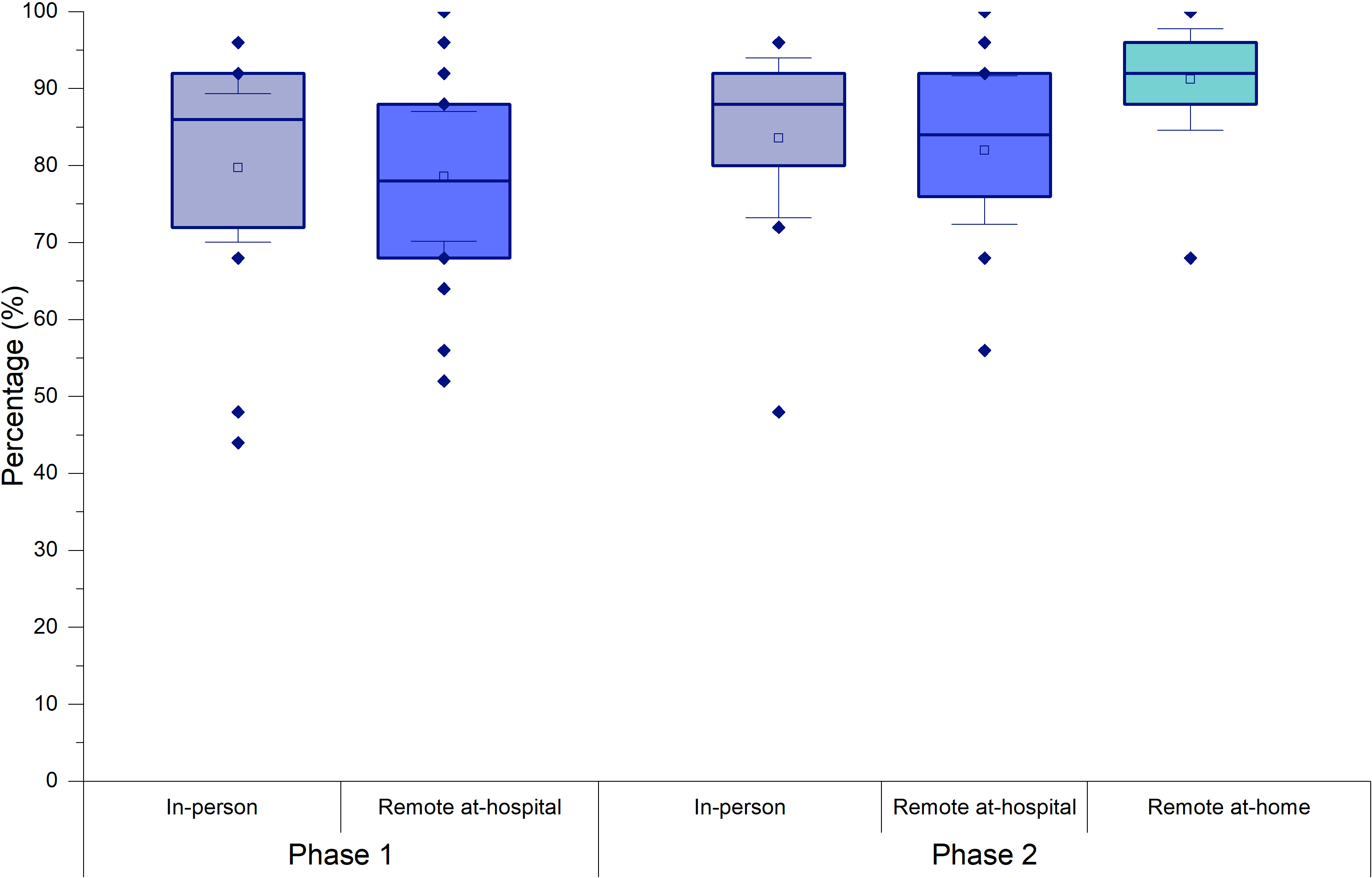
For the audio-only monitored live voice bisyllabic word tests carried out in the sound booth, there was no significant difference between the percentage of correct words with the in-person and remote-at-hospital programs in phase 1 (n = 14; z = 0.140; p = 0.914; rrb = 0.044). There was also no difference between word recognition for the in-person program, the remote program at the hospital, and the remote program at home in phase 2 (n = 10;X²F (2) = 3.500; gl = 2; p = 0.174; W = 0.175) (Figure 3).
Percentage of correct words on the monitored live voice bisyllabic word test (audio-only) in the sound booth in phases 1 and 2.
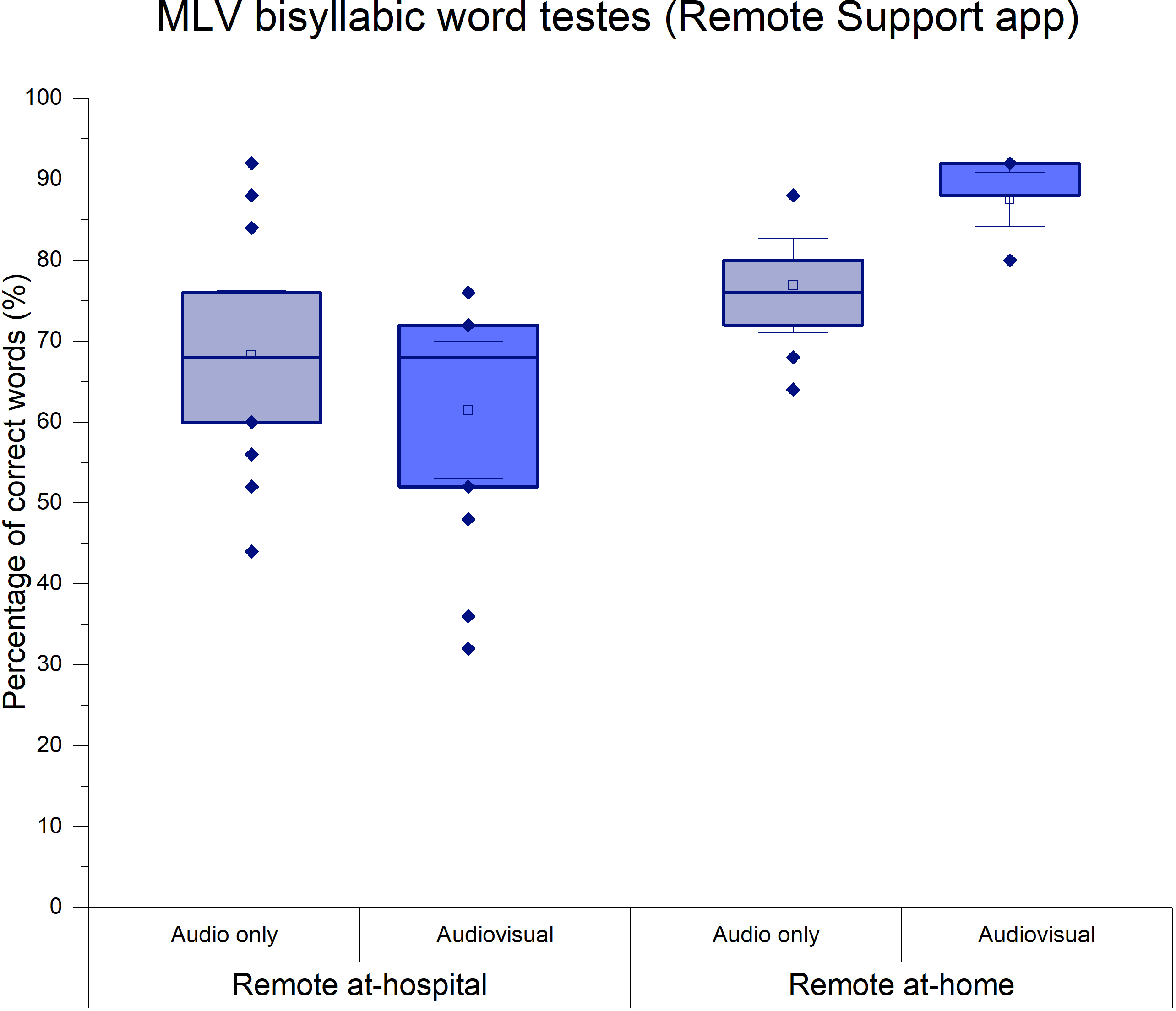
Bisyllabic word testing with monitored live voice was also feasible via the remote programming app, with and without visual cues, at both remote programming locations. There was a significant difference in the percentage of correct words between the two remote programming locations (n = 10; X²F (2) = 14.172; p = 0.003*; W = 0.525). No significant difference was observed in the test with audio-only, carried out at home or in hospital (n = 10; t = 1.374; p = 0.182; rrb = -0.472). However, the percentage of correct word scores with audiovisual cues was higher at home (mean = 87.56%; SD = 4.67; 95% CI = 83.97 to 91.14) than in-hospital sessions (mean = 73.78%; SD = 12.02; 95% CI = 64.54 to 83.02), (n = 10; t = 2.656; p = 0.014*; rrb = -0.889). There was also a significant improvement with access to visual cues at home (n = 10; t = 2.198; p = 0.038*; rrb = -0.722), (mean = 87.6%; SD = 4.67; 95% CI = 83.97 to 91.14) than those with audio only (mean = 76.9%; SD = 8.19; 95% CI = 70.59 to 83.19) (Figure 4).
Percentage of correct words on the MLV bisyllabic word test (audiovisual and audio-only) measured via the remote support app in phases 1 and 2.
Source: Prepared by the authors based on the results of the study.
Cochlear implants recipient feedback on remote programming experience and acceptability
Figure 5 shows the average ratings participants provided for statements related to remote programming after each experience in both remote sessions (at-hospital [n = 14] and at-home [n = 10]). On average, participants agreed or strongly agreed with most statements regarding communication during the remote session, ease of use, effectiveness, and relevance to future implant care. There were no significant differences in ratings between in-hospital and remote at-home programming across any of the questions (p > 0.05). The ratings tended to be higher in the remote at-home condition for most statements (FIGURE 5).
Participants' average subjective ratings on their experience with remote programming during both remote sessions (at-hospital and at-home).
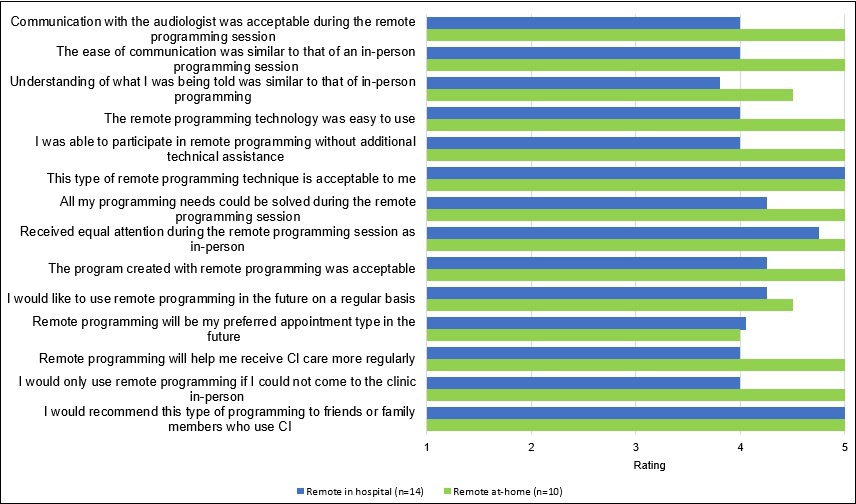
Source: Prepared by the authors based on the results of the study.
Figure 6 presents the individual Mobile Device Proficiency Questionnaire-16 (MPDQ-16) scores, perceived ease of use of the remote programming technology, need for additional technical assistance, and the percentage of correct words on the word test obtained via the app with audio only.
Individual MPDQ-16 scores (shown in green), perceived ease of use of the remote programming technology (light blue), need for additional technical assistance (dark blue), and percentage of correct words on the monitored live voice word test using the app through audio-only (represented by the black dot).
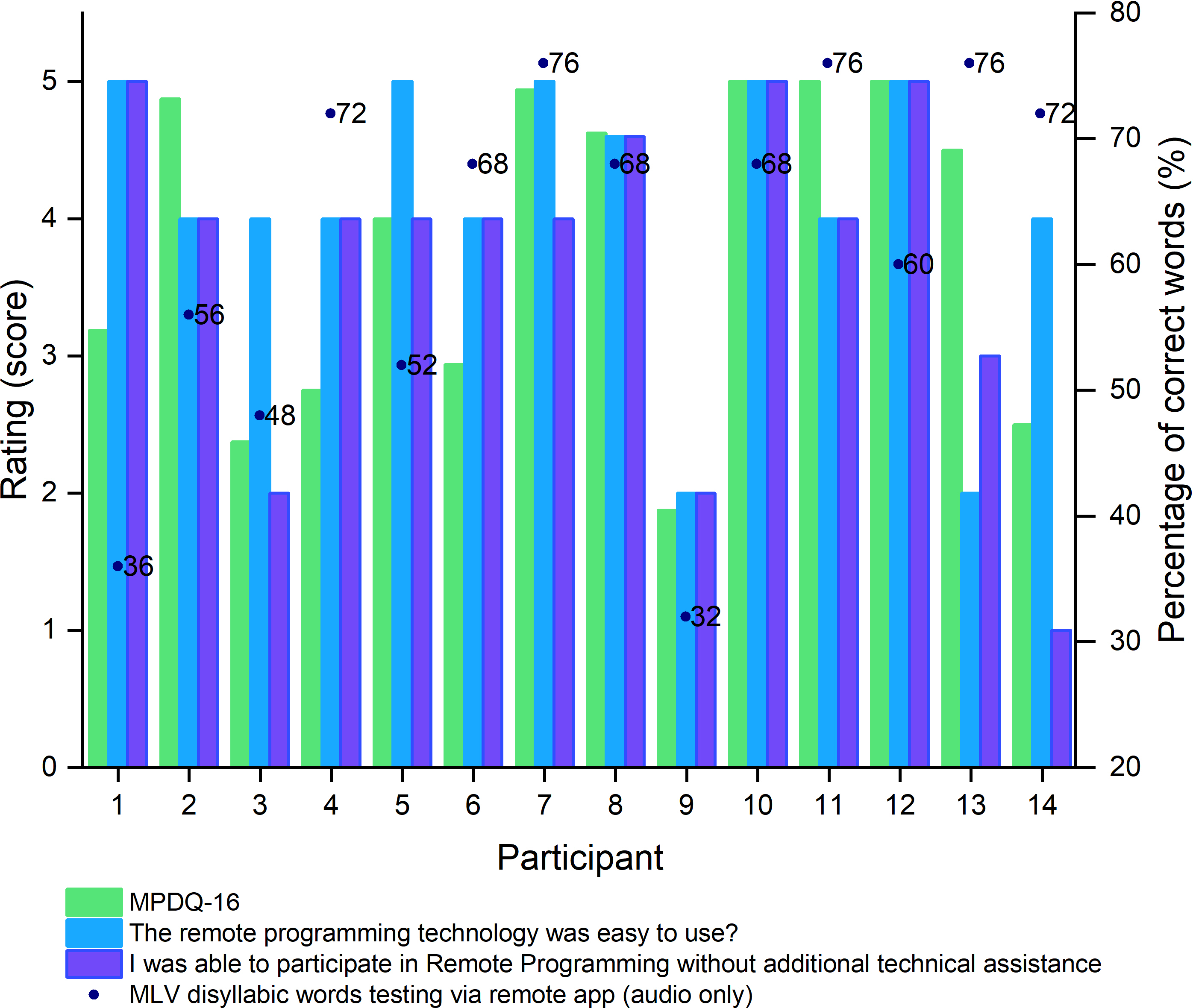
Source: Prepared by the authors based on the results of the study.
Most participants perceived the duration of the in-person programming sessions to be similar to remote programming carried out in hospital (78.6%), at home (50.0%), or at a hospital (78.6%). Half of the participants lived in the metropolitan area and the main benefits cited included: time savings (n = 8; 57%), convenience (n = 7; 50.0%), less, or no travel required (n = 5; 35.7%), reduced travel expenses (n = 5; 35.7%) and reduced loss of work, or school time (n = 5; 35.7%).
Audiologist’s feedback on remote programming experience and utility
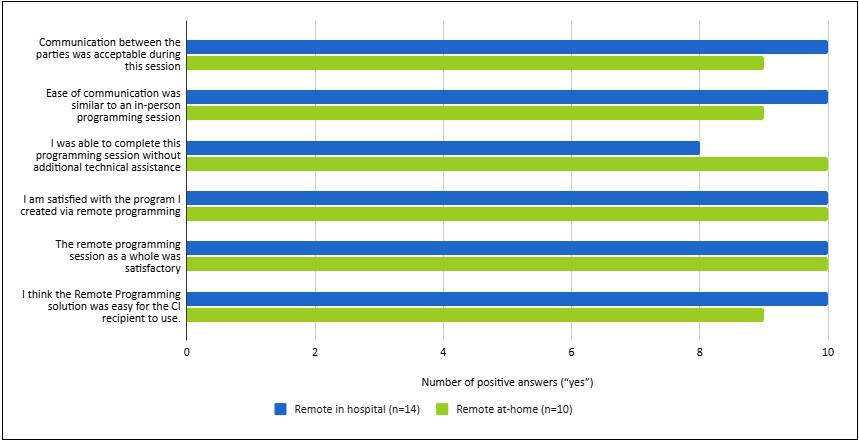
Figure 7 presents the results from the per-remote-programming-session questionnaire completed by the audiologists regarding the remote-programming experience of the 10 subjects who participated in both phases of the study. Number of positive responses (“yes” vs “no”) ranged between 8 and 10 per question.
Number of positive answers by the audiologist on the final questionnaire for both remote sessions (in hospital and at-home).
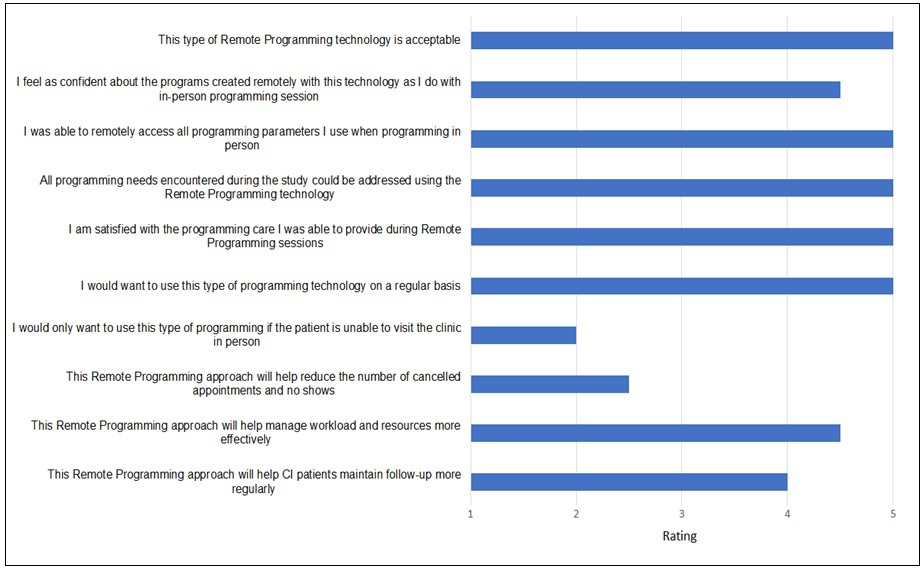
Results from the end-of-the study questionnaire indicated that the two audiologists agreed or strongly agreed (rated on average between 4.5 and 5 points) to statements regarding acceptability, ease, reliability, and efficiency (Figure 8).
Audiologist’s subjective rating average on the final questionnaire (N= 2).
Discussion
This study evaluated the usability, effectiveness, and acceptability of remote cochlear implant programming in adult recipients and examined whether outcomes were comparable to those obtained during conventional in-person sessions. Overall, the findings indicate that remote programming, conducted both in-hospital and at home, can achieve clinical outcomes equivalent to in-person care and is well accepted by both recipients and audiologists.
Across objective measures of speech perception, maps created remotely were noninferior to in-person maps in sentence comprehension, suggesting that remote access to programming parameters and objective measures is sufficient to maintain speech understanding in experienced implant users. The slightly higher sentence recognition observed during the second phase of the study could be explained by increased familiarity with the testing procedures and confidence in the technology, rather than by any intrinsic advantage of remote programming itself. Together, these findings support the interpretation that remote programming does not compromise auditory performance.
The absence of differences in word recognition scores obtained in the sound booth further reinforces the clinical equivalence between remote and in-person programming. These results are consistent with previous reports demonstrating comparable outcomes between remote and in-person cochlear implant programming across a range of measures, including pure-tone thresholds [11,12,22,23], speech reception thresholds [12], monosyllabic word recognition in quiet and in noise [5,10,12,22,23], sentence recognition in quiet conditions [11,14,22] and perceived auditory functioning [23], even when stimulation levels differed between mappings [22].
An important and novel observation emerged from the bisyllabic word testing conducted via the remote programming application. Participants achieved higher scores during at-home remote sessions when audiovisual cues were available, whereas this effect was not observed in the in-hospital remote condition. Earlier studies identified delayed signal transmission and audiovisual asynchrony as challenges during remote care [5,12,24]. In contrast, although internet speed did not differ significantly across settings in the present study, higher average home speeds may have improved video quality and audiovisual synchrony, thereby enhancing access to orofacial cues and supporting better speech comprehension with visual input.
Environmental noise has also been described as a potential limitation of remote programming [25]. However, the comparable ambient noise levels observed across in-person, in-hospital remote, and at-home remote sessions in the present study indicate that environmental noise did not meaningfully influence speech perception outcomes. This suggests that, when basic acoustic conditions are ensured, remote programming can be conducted in diverse environments without compromising auditory assessment.
User-related factors played a central role in interpreting the findings. Although smartphone proficiency varied across participants, most cochlear implant recipients rated the remote programming application as easy to use and highly acceptable. Importantly, individuals with lower self-reported digital proficiency but greater geographic distance from the specialized centers still opted for at-home remote programming. This pattern suggests that perceived benefits, such as reduced travel time, cost savings, and convenience, may outweigh concerns related to technological complexity, highlighting the potential of remote programming to mitigate access barriers for geographically remote patients.
From a clinical perspective, the consistently positive subjective ratings regarding communication quality, attention from the audiologist, and overall satisfaction indicate that remote programming can preserve the therapeutic relationship to cochlear implant care. Participants generally perceived remote sessions as comparable to in-person visits and expressed willingness to use this modality in the future, in line with earlier studies reporting high acceptability and satisfaction with remote care [5,10,11,14,23,24,25]. Slightly higher ratings for at-home sessions may reflect greater comfort with the home environment and increased familiarity with the remote programming process.
Audiologists’ feedback further supports the clinical viability of remote programming. Clinicians reported no substantial difficulties in communication, application handling, or access to necessary programming tools, and they consistently rated the sessions as acceptable, reliable, and efficient. These observations corroborate previous findings indicating positive professional acceptance of remote implant programming and its equivalence to in-person care from the provider perspective [5,10,14,23,24,26].
Taken together, the present findings align with and extend earlier research demonstrating that remote cochlear implant programming is safe [10,11], practical, and effective compared to in-person sessions [14,22,23]. By reducing travel burden and associated costs, remote programming may offer a cost-effective solution, particularly for individuals with limited mobility or those residing far from specialized centers [11], thereby contributing to more equitable access to specialized hearing healthcare services.
Despite these strengths, some limitations must be acknowledged. The sample size was small, and participant withdrawal limited the scope of statistical analyses. In addition, the study population consisted predominantly of experienced cochlear implant users with relatively good speech recognition, which may restrict the generalizability of the findings. Future studies with larger samples are warranted to determine the effectiveness of this technology among new users and among individuals with poorer speech-recognition performance. Further research should also explore the relationship between speech recognition outcomes and the need for in-person support during remote implant programming, as well as assess long-term outcomes and sustained adoption of this model of care.
Conclusions
Remote programming can be successfully used for cochlear implant follow-up in Chile. Its efficacy was comparable to in-person programming, as evidenced by equivalent speech-perception scores and satisfaction ratings reported by cochlear implant recipients. The ability to provide standard remote programming care in real-time has the potential to significantly reduce the burden of in-person visits and increase equity in access to implant follow-up.

