Evaluaciones económicas
← vista completaPublicado el 29 de junio de 2018 | http://doi.org/10.5867/medwave.2018.03.7220
Análisis costo-efectividad de lenalidomida en combinación con dexametasona frente a bortezomib combinado con dexametasona en el tratamiento en segunda línea del mieloma múltiple en Chile
Cost-effectiveness of lenalidomide in combination with dexamethasone compared to bortezomib in combination with dexamethasone for the second-line treatment of multiple myeloma in Chile
Resumen
CONTEXTO El mieloma múltiple es una neoplasia de las células plasmáticas de la medula ósea. Las terapias disponibles no son curativas y la mayoría de los pacientes se vuelve refractario al tratamiento. Agentes como lenalidomida y bortezomib han demostrado su eficacia en el tratamien-to en segunda línea de estos pacientes.
OBJETIVO Evaluar el costo-efectividad de la combinación lenalidomida/dexametasona frente a bortezomib/dexametasona en pacientes con mieloma múltiple, no candidatos a trasplante, previamente tratados con bortezomib, desde la perspectiva del sistema nacional de salud chileno.
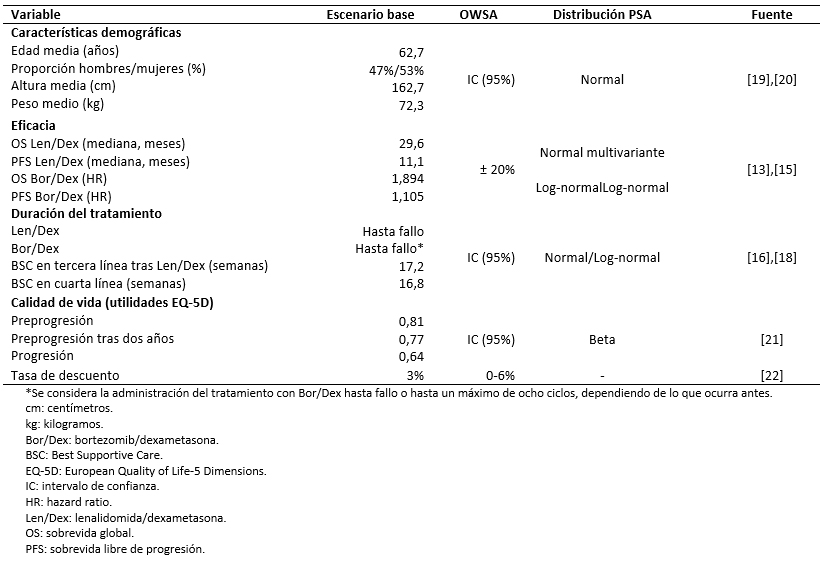
METODOLOGÍA Se empleó un modelo de Markov que simula la evolución de una cohorte de pacientes a través de cuatro estados de salud (preprogresión en tratamiento, preprogresión sin tratamiento, progresión o muerte) en un horizonte temporal de 25 años. Los datos de eficacia, uso de recursos y frecuencia de efectos adversos fueron extraídos de los ensayos sobre mieloma múltiple MM-009 y MM-010 y de un estudio retrospectivo de retratamiento con bortezomib. Todos los parámetros fueron validados por expertos. Se aplicó una tasa de descuento en costos y beneficios de 3%. La robustez de los resultados fue evaluada mediante un análisis de sensibilidad univariante y probabilístico.
RESULTADOS El tratamiento con lenalidomida/dexametasona proporciona 1,41 años de vida y 0,83 años de vida ajustados por calidad incrementales respecto a bortezomib/dexametasona, con un costo incremental de 11 864 597,86 pesos chilenos (19 589,86 dólares). La ratio de cos-to-efectividad y costo-utilidad incremental se cifró en 8 410 266,92 pesos chilenos (13 886,35 dólares) por año de vida ganado y 14 271 896,16 pesos chilenos (23 564,59 dólares) por año de vida ajustado por calidad respectivamente.
CONCLUSIÓN La lenalidomida/dexametasona representa una alternativa potencialmente costo-efectiva, desde la perspectiva del sistema nacional de salud chileno, para el tratamiento en segunda línea de pacientes con mieloma múltiple no candidatos a trasplante.
Introducción
El mieloma múltiple es una neoplasia maligna que se caracteriza por la proliferación clonal de células plasmáticas originadas de células B de la médula ósea [1]. Generalmente va precedido de una fase pre-maligna asintomática denominada gammapatía monoclonal de significación indeterminada, que puede evolucionar directamente a mieloma múltiple sintomático o progresar a un estadio intermedio, el mieloma quiescente. En este último caso, se estima que el 10% progresará a mieloma múltiple sintomático durante los primeros cinco años [2]. La evolución de la enfermedad va asociada a un deterioro significativo de la calidad de vida, debido principalmente a las complicaciones asociadas. Entre ellas, las más relevantes incluyen anemia, trombocitopenia, leucopenia, fractura ósea, hipercalcemia e insuficiencia renal [1],[3].
Pese a los avances en el tratamiento del mieloma múltiple, sigue observándose una elevada tasa de mortalidad en estos pacientes, registrándose más de 260 000 fallecimientos al año por esta causa a nivel global [4]. En Chile, se estima una incidencia de 2,3 casos por 100 000 habitantes por año y una prevalencia a cinco años del 1% [4].
En ausencia de una terapia definitiva [5], la introducción de agentes como la talidomida, lenalidomida y bortezomib ha permitido aumentar sensiblemente la sobrevida de los pacientes con mieloma múltiple. La selección del tratamiento adecuado depende del riesgo del paciente y de si este es elegible para el trasplante autólogo de células madre [6],[7].
En pacientes no candidatos a trasplante, se recomienda un esquema de tres o dos medicamentos, dependiendo de su estado general, basado en bortezomib, lenalidomida o talidomida [6],[7],[8]. En caso de recaída o progresión, las principales guías de práctica clínica recomiendan repetir el mismo tratamiento o considerar el cambio a otra terapia (lenalidomida/dexametasona o bortezomib/doxorubicina pegilada) [6],[7],[8].
En este contexto, el objetivo de este trabajo es estimar la razón de costo-efectividad incremental del tratamiento en segunda línea con lenalidomida en combinación con dexametasona, frente al retratamiento con bortezomib combinado con dexametasona en pacientes con mieloma múltiple y tratamiento previo con bortezomib, desde la perspectiva del ámbito sanitario público chileno.
Métodos
Para llevar a cabo el objetivo propuesto, se adaptó al ámbito sanitario chileno un modelo que se presentó originariamente en el National Institute for Health Care and Excellence (NICE) en su adaptación al Reino Unido y que estima los beneficios clínicos y costos de lenalidomida en combinación con dexametasona frente a bortezomib combinado con dexametasona en el tratamiento de pacientes con mieloma múltiple no candidatos a trasplante, previamente tratados con bortezomib [9].
Los parámetros empleados en el modelo fueron validados por dos hematólogos expertos en el tratamiento del mieloma múltiple en Chile (CP y CG). Para la elaboración del presente trabajo se han utilizado las directrices del Consolidated Health Economic Evaluation Reporting Standards (CHEERS) [10].
Estructura del modelo
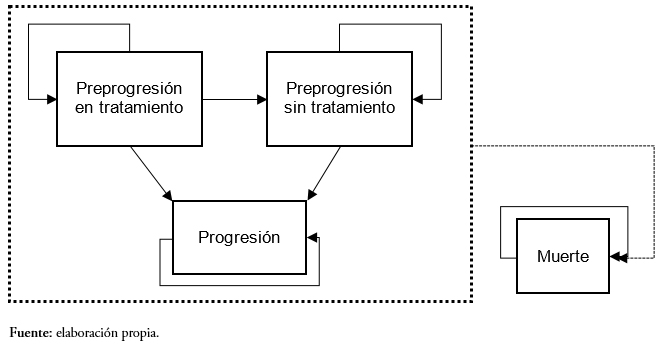
El diseño es un modelo de Markov que simula la evolución de una cohorte hipotética de pacientes desde la asignación del tratamiento hasta finalizar el horizonte temporal (25 años). De acuerdo a las recomendaciones de la Sociedad Internacional de Farmacoeconomía e Investigación de Resultados (ISPOR, por su sigla en inglés International Society For Pharmacoeconomics and Outcomes Research), la elección de este tipo de diseño resulta adecuada si se considera la extensión del horizonte temporal y a la naturaleza de la enfermedad, que implica que los pacientes puedan encontrase en distintas fases de la enfermedad o estados de salud [11]. Asimismo, los modelos de Markov son apropiados cuando el análisis requiere la simulación de varias líneas de tratamiento consecutivas [12].
Desde su entrada en el modelo, los pacientes transitan a través de cuatro posibles estados de salud (Figura 1). Inicialmente todos los pacientes se encuentran en el estado preprogresión en tratamiento, que corresponde a pacientes estables que están recibiendo tratamiento (lenalidomida en combinación con dexametasona o bortezomib junto con dexametasona). A partir de ese estado los pacientes pueden permanecer en su estado o transitar a uno de los siguientes estados:
- Preprogresión sin tratamiento: pacientes sin tratamiento, es el estado donde se encuentran quienes han experimentado fallo del tratamiento y que, por tanto, pasarán a recibir la siguiente línea de tratamiento;
- Progresión: basado en lo establecido en los ensayos sobre mieloma múltiple MM-009 y MM-010. En este estado se encuentran aquellos pacientes que muestran signos de enfermedad progresiva definida como un aumento de al menos 25% en la proteína M a partir del valor mínimo; un aumento absoluto en la proteína M sérica de más de 500 miligramos por decilitro, en comparación con el valor mínimo; un aumento absoluto en la proteína M urinaria de más de 200 miligramos por un período de 24 horas; o bien una nueva lesión ósea, plasmacitoma (o un aumento en el tamaño de tales lesiones), o un nivel de calcio sérico de más de 11,5 miligramos por decilitro [13],[14]
- Muerte.
Las probabilidades de transición a los estados de salud de preprogresión sin tratamiento, progresión y muerte se han obtenido a partir de las curvas de sobrevida libre de progresión, tiempo hasta el fallo y sobrevida global respectivamente, de los estudios sobre mieloma múltiple MM-009 y MM-010, en los que se evaluó la eficacia de lenalidomida en combinación con dexametasona frente a placebo en conjunto con dexametasona en pacientes previamente tratados, y del estudio retrospectivo de retratamiento con bortezomib [13],[14],[15].
 Tamaño completo
Tamaño completo Tratamientos comparados
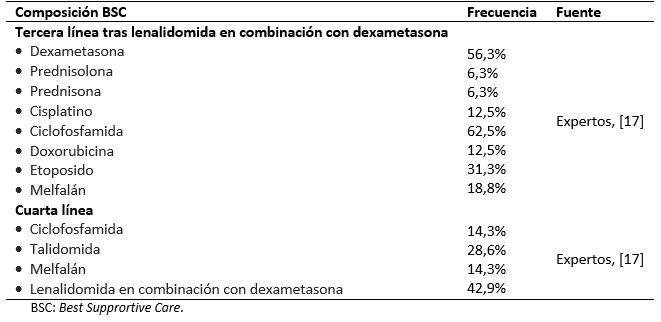
El presente análisis compara los resultados asociados al tratamiento en segunda línea de lenalidomida en combinación con dexametasona frente al retratamiento con bortezomib junto con dexametasona. El modelo asume que todos los pacientes han recibido una primera línea de tratamiento con bortezomib. Tras su entrada en el modelo, los pacientes son asignados a recibir lenalidomida en combinación con dexametasona o retratamiento con bortezomib en conjunto con dexametasona hasta fallo (el tratamiento en segunda línea con bortezomib en combinación con dexametasona es limitado a un máximo de ocho ciclos según indicado en la ficha técnica) [16]. En tercera línea, los pacientes tratados con bortezomib en combinación con dexametasona pasan a recibir lenalidomida en combinación con dexametasona, mientras que los pacientes tratados con lenalidomida combinado con dexametasona reciben Best Supportive Care, un conjunto de tratamientos formado por quimioterápicos (ciclofosfamida, melfalán, cisplatino, doxorrubicina, etoposido) y eventualmente lenalidomida (Tabla 1) [17],[18]. En cuarta línea de tratamiento ambos brazos reciben el Best supportive Care.
 Tamaño completo
Tamaño completo Población
El tratamiento de primera línea en pacientes con mieloma múltiple no candidatos a trasplante consiste en esquemas de tres o dos medicamentos basados en bortezomib, lenalidomida o talidomida. En caso de recaída, se recomienda repetir el mismo tratamiento o considerar el cambio a otra terapia (lenalidomida/dexametasona o bortezomib/doxorubicina pegilada) [6],[7],[8]. Sobre esta base, la población considerada en el presente análisis corresponde a pacientes con mieloma múltiple no candidatos a trasplante previamente tratados con bortezomib. Las características sociodemográficas y clínicas de la población fueron obtenidas de los ensayos clínicos sobre mieloma múltiple MM-009 y MM-010 [13],[14]. No obstante, con el objetivo de ajustar las curvas de sobrevida y el cálculo del área de superficie corporal a la población chilena, la proporción de hombres/mujeres con mieloma múltiple, el peso y la altura medios fueron extraídos de la literatura (Tabla 2) [19],[20].
 Tamaño completo
Tamaño completo Horizonte temporal y perspectiva del estudio
Se definieron ciclos de 28 días, en concordancia con la duración de los ciclos de los tratamientos principales, según lo indicado en ficha técnica. Esta elección está en línea con las recomendaciones de la Sociedad Internacional de Farmacoeconomía e Investigación de Resultados, que indican que “la duración de los ciclos del modelo debe ser lo suficientemente corta como para representar la frecuencia de los eventos clínicos y de las intervenciones” [11]. En total, el modelo se compone de 326 ciclos que corresponden a un horizonte temporal de 25 años, lo suficientemente largo como para capturar los costos y efectos clínicos relevantes.
El sistema de salud chileno consta de dos sectores: el público, constituido principalmente por el Fondo Nacional de Salud; y el privado, conformado principalmente por los seguros de salud llamados instituciones de salud previsional [24]. En el presente análisis, se adoptó la perspectiva del sistema nacional de salud público chileno.
Tasa de descuento
Se aplicó una tasa de descuento en costos y beneficios del 3% [22].
Eficacia
El modelo de Markov empleado en el presente estudio utiliza las probabilidades de transición entre los distintos estados de salud para estimar los años de vida ganados a lo largo del horizonte temporal. Estas probabilidades se obtienen de las curvas de sobrevida global y sobrevida libre de progresión de los tratamientos. Se llevó a cabo una revisión sistemática de la literatura con el objetivo de identificar la evidencia disponible relativa a la eficacia de los tratamientos evaluados [25]. Las curvas de lenalidomida en combinación con dexametasona han sido modelizadas a partir de los resultados agregados de los estudios sobre mieloma múltiple MM-009 y MM-010 y parametrizados mediante distribuciones Gompertz (sobrevida global) y Γ (sobrevida libre de progresión) [13],[14],[15]. Dada la falta de una comparación directa entre bortezomib en combinación con dexametasona y lenalidomida junto con dexametasona, las curvas de sobrevida global y sobrevida libre de progresión de bortezomib combinado con dexametasona se han reconstruido a partir del hazard ratio de la comparación indirecta entre los ensayos sobre mieloma múltiple MM-009 y MM-010 y el estudio retrospectivo de retratamiento con bortezomib [13],[14],[15]. Los hazard ratio se han obtenido como la razón entre la mediana de sobrevida de ambos tratamientos, tras ajustar la eficacia de lenalidomida en combinación con dexametasona a las características de la población del estudio comparador (Tabla 2).
El presente modelo estima también los años de vida ajustados por calidad como la suma de los años de vida transcurridos en cada estado de salud, ponderados por la utilidad asociada. Los datos de utilidad, así como los de disutilidad asociada a los eventos adversos fueron extraídos de la literatura [22],[26],[27],[28],[29],[30].
Costos
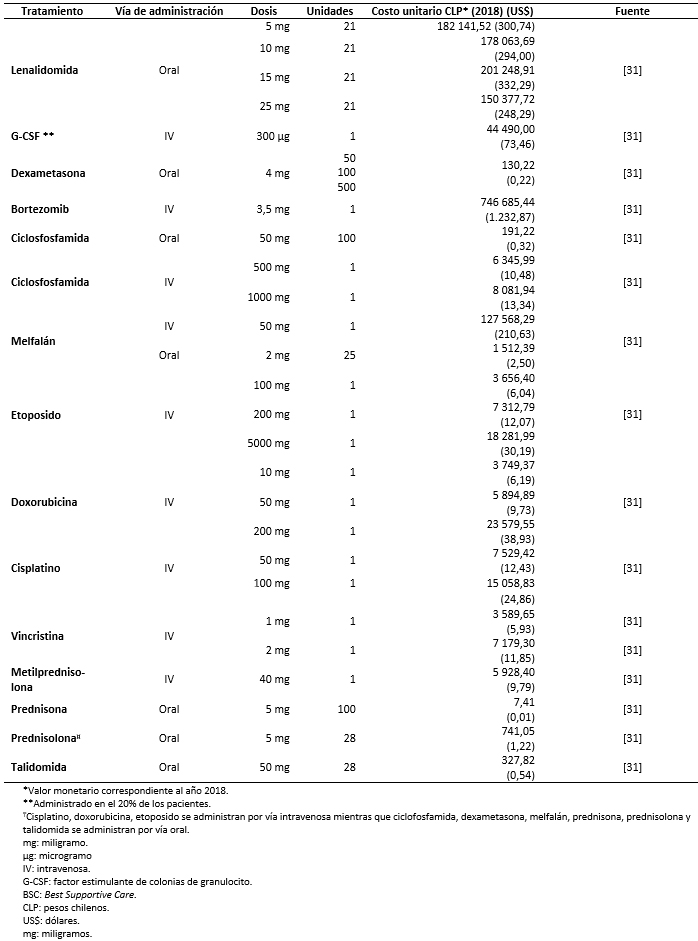
Los costos totales fueron estimados como la suma de costos farmacológicos (Tabla 3), costos asociados al uso de recursos (Tabla 4) y manejo de eventos adversos (Tabla 5) [16],[17],[18],[31],[32],[33],[34],[35]. Los costos asociados al uso de recursos incluyen costos asociados a pruebas de seguimiento y monitorización, administración, consultas y cuidados paliativos (Tabla 6).
 Tamaño completo
Tamaño completo 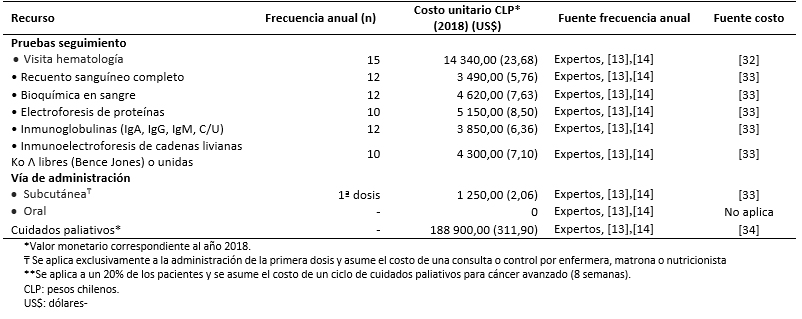 Tamaño completo
Tamaño completo 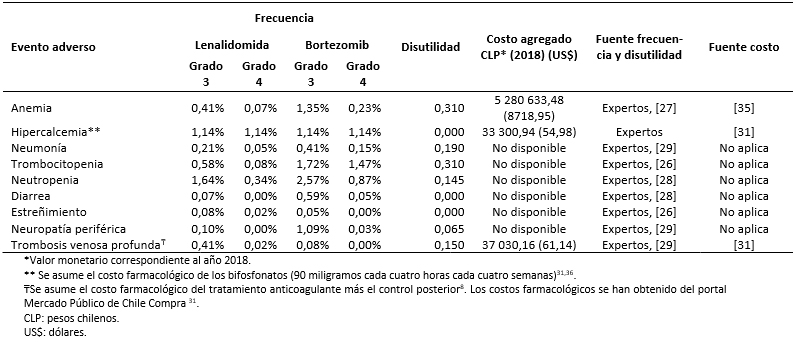 Tamaño completo
Tamaño completo 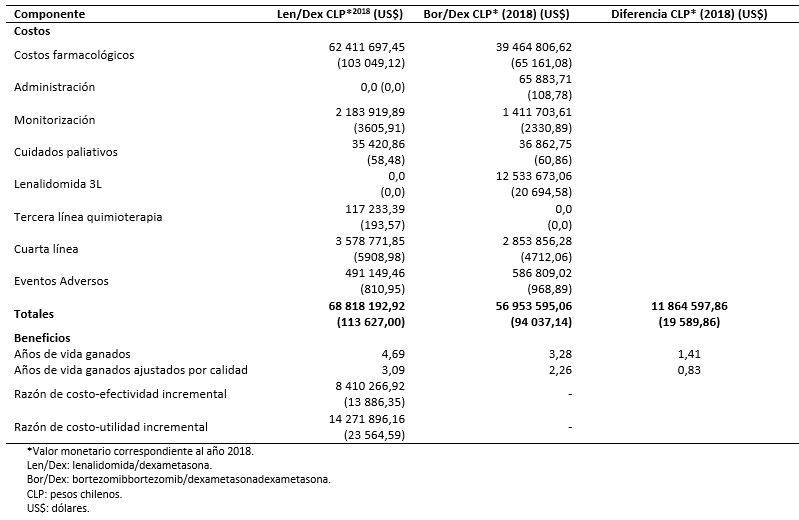 Tamaño completo
Tamaño completo Todos los costos fueron estimados en pesos chilenos y actualizados a 2018. Los valores de cambio, y conversión fueron tomados desde el Banco Central de Chile en el día 28 de marzo de 2018 [37]. Los costos farmacológicos fueron obtenidos de la base de datos del portal Mercado Público de Chile Compra [31]. Para cada uno de los medicamentos considerados se obtuvieron los costos de distintas licitaciones y se calculó un promedio por miligramo. Las dosis y frecuencia de administración fueron obtenidas de las fichas técnicas y ajustadas según recomendaciones de los expertos. Los costos de uso de recurso fueron extraídos de los aranceles del Fondo Nacional de Salud (2018) [32],[33]. La frecuencia de uso de recursos fue obtenida de los estudios sobre mieloma múltiple MM-009 y MM-010 y validada por expertos [13],[14].
Variables de resultado
A partir de los años de vida ganados, los años de vida ganados ajustados por calidad y los costos totales acumulados por el paciente, se han estimado la razón de costo-efectividad incremental y costo-utilidad incremental como el cociente entre el costo incremental de lenalidomida en combinación con dexametasona frente a bortezomib junto con dexametasona, y la diferencia en años de vida o años de vida ajustados por calidad ganados..
Análisis de sensibilidad
Se realizó un análisis de sensibilidad univariante y un análisis de sensibilidad probabilístico para evaluar la robustez de los resultados bajo la incertidumbre o variabilidad de los parámetros del modelo. El análisis de sensibilidad univariante muestra los resultados obtenidos al modificar, de manera individual, cada uno de los parámetros entre un valor mínimo y máximo establecidos. Estos valores se han obtenido a partir del intervalo de confianza al 95% o introduciendo una variación del ± 20% sobre el valor del caso base. Para la tasa de descuento, el análisis de sensibilidad univariante contempla una variación entre 0% y 6% según recomendaciones de la guía de evaluaciones económicas del Ministerio de Salud de Chile [22]. El análisis de sensibilidad probabilístico se ha implementado mediante una simulación de Montecarlo de 1000 iteraciones, mediante la cual se asignan valores aleatorios a cada uno de los parámetros del modelo según su propia distribución de probabilidad (Tabla 2).
Resultados
En la Tabla 6 se muestran los beneficios y los costos asociados a las dos cohortes de tratamiento. A lo largo del horizonte temporal, los pacientes asignados al tratamiento con lenalidomida en combinación con dexametasona acumulan 4,69 años de vida (3,09 años de vida ajustados por calidad) frente a 3,28 años de vida (2,26 años de vida ajustados por calidad) del tratamiento con bortezomib en combinación con dexametasona. El costo del tratamiento con lenalidomida combinado con dexametasona se cifra en 68 818 192,92 pesos chilenos (113 627,00 dólares) frente a 56 953 595,06 pesos chilenos (94 037,14 dólares) del comparador. En la Tabla 6 se desglosan los costos acumulados por los dos brazos de tratamiento a lo largo de la simulación. Los resultados muestran que lenalidomida en combinación con dexametasona sería más eficaz y más costoso respecto a bortezomib en combinación con dexametasona, aportando 1,41 años de vida (0,83 años de vida ajustados por calidad) incrementales y asociándose a un costo incremental de 11 864 597,86 pesos chilenos (19 589,86 dólares) (Tabla 6).
La Organización Mundial de la Salud [38] sugiere considerar como muy costo-efectivas las intervenciones cuya razón de costo-efectividad incremental sea inferior a un Producto Interno Bruto (PIB) per cápita y como “no costo efectivas” aquellas intervenciones que superen los tres PIB per cápita (en este caso 9 136 835,90 pesos chilenos equivalentes a 15 086 dólares - 27 410 507,70 pesos chilenos equivalentes a 45 258 dólares) por año de vida ganado ajustado por calidad [39]. Sobre esta base, el tratamiento con lenalidomida en combinación con dexametasona podría considerarse como potencialmente costo-efectivo frente al retratamiento con bortezomib asociándose a una razón de costo-efectividad incremental inferior a un PIB per cápita (8 410 266,92 pesos chilenos equivalentes a 13 886,35 dólares por año de vida ganado) y de costo-utilidad incremental inferior a tres veces el PIB (14 271 896,16 pesos chilenos equivalentes a 23 564,59 dólares por año de vida ajustado por calidad adicional).
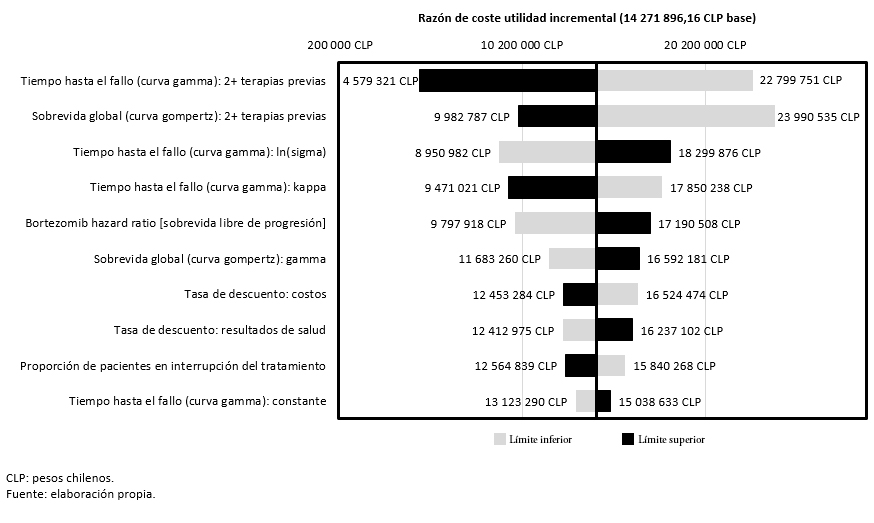
El análisis de sensibilidad univariante muestra que las variables con mayor impacto en los resultados del análisis son el tiempo hasta el fallo y la sobrevida global en pacientes que hayan recibido dos o más terapias previas. En concreto, un incremento o un decremento del 20% en el tiempo hasta el fallo, haría oscilar el costo-utilidad incremental (valor basal) entre 4 579 321 pesos chilenos (7 561,00 dólares) y 22 799 751 pesos chilenos (37 645,09 dólares), respectivamente. Por otra parte, una variación de ± 20% en la sobrevida global se traduciría en una oscilación del costo-utilidad incremental comprendida entre 9 982 787 pesos chilenos (16 482,77 dólares) y 23 990 535 pesos chilenos (39 611,22 dólares) (Figura 2).
Los resultados del análisis de sensibilidad probabilístico muestran que en el 98,4% de las simulaciones, el tratamiento con lenalidomida en combinación con dexametasona sería potencialmente costo-efectivo frente a bortezomib junto con dexametasona, asociándose a un costo-utilidad incremental inferior al umbral de 27 410 507,70 pesos chilenos (45 258 dólares) (Figura 3).
 Tamaño completo
Tamaño completo 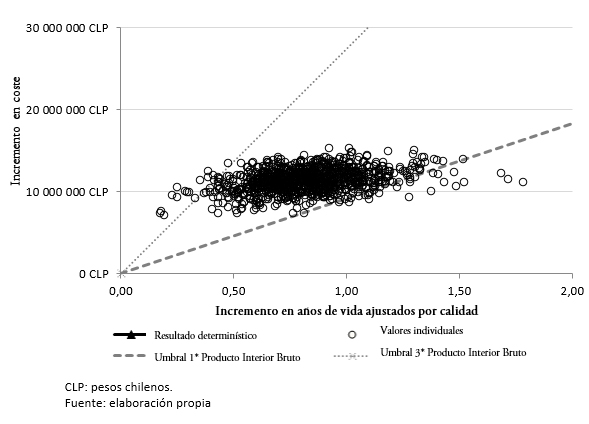 Tamaño completo
Tamaño completo Discusión
El análisis de los costos y beneficios asociados a las alternativas actualmente disponibles para el tratamiento del mieloma múltiple es esencial para asegurar una asignación eficiente de los recursos. El presente estudio muestra que en el ámbito sanitario chileno, la lenalidomida en combinación con dexametasona sería una alternativa potencialmente costo-efectiva en el tratamiento en segunda línea de pacientes con mieloma múltiple no candidatos a trasplante, aportando un incremento significativo de la sobrevida sin superar el umbral de tres veces el PIB per cápita por año de vida ganado ajustado por calidad [38].
Los resultados hallados concuerdan con los reportados por otros estudios que han evaluado el costo-efectividad de la lenalidomida en combinación con dexametasona en el mismo grupo de pacientes, en otros entornos [40],[41]. En un estudio realizado en Noruega, el tratamiento con lenalidomida en combinación con dexametasona resultó costo-efectivo frente a bortezomib, asociándose a un mayor número de años de vida (4,06 versus 3,11) y a años de vida ajustados por calidad (2,95 versus 2,19) y a un costo incremental de 188 245 coronas noruegas (valor monetario a 2011), estimándose una razón de costo-efectividad de 198 714 coronas noruegas (valor monetario a 2011) por año de vida y un costo-utilidad incremental de 247 078 coronas noruegas (valor monetario a 2011) por año de vida ajustados por calidad [40].
De modo similar, en un estudio llevado a cabo en el ámbito sanitario griego, se estimó un incremento de 0,99 años de vida y 0,79 años de vida ajustados por calidad a favor de lenalidomida en combinación con dexametasona y un costo incremental de 28 741 euros, calculándose una razón de costo-efectividad incremental y de coste utilidad incremental de 29 415 euros por año de vida y de 38 268 euros por año de vida ajustado por calidad respectivamente [41]. Sobre la base de estos resultados, los autores concluyeron que el tratamiento con lenalidomida en combinación con dexametasona sería coste-efectivo frente a bortezomib, teniendo en cuenta una disposición a pagar tres veces el Producto Interno Bruto per cápita en Grecia (60 000 euros -según valor monetario a 2013- por año de vida ajustado por calidad).
Cabe destacar que contrariamente al presente estudio, los datos relativos a la eficacia de bortezomib empleados en los análisis económicos noruego y griego fueron extraídos del ensayo clínico APEX en el que se comparó la eficacia de bortezomib y dexametasona en altas dosis en pacientes con mieloma múltiple previamente tratados [42]. No obstante, en el estudio APEX, el tratamiento previo con bortezomib constituía un criterio de exclusión del ensayo, por lo que en el presente análisis no se consideró oportuno emplear esa fuente ya que la población incluida en ese estudio difiere sustancialmente de la población de interés (pacientes previamente tratados con bortezomib).
Por otra parte, en un estudio llevado a cabo en el entorno sueco, se concluyó que el tratamiento con bortezomib sería dominante frente a lenalidomida en combinación con dexametasona, asociándose a un mayor número de años de vida ajustados por calidad (2,95 versus 2,91 meses) y generando un ahorro de 546 126 coronas suecas (según el valor monetario de 2010) [43]. La divergencia entre estos resultados y los del presente trabajo se debe probablemente a diferencias metodológicas entre los mencionados estudios [43]. En concreto, el análisis sueco contempla solo tres posibles estados de salud (preprogresión, progresión y muerte) y utiliza un método paramétrico de modelización de la sobrevida (partitioned survival model) en el que la proporción de pacientes en cada estado es calculada en base a ecuaciones paramétricas [43].
En el presente análisis, por el contrario, se eligió emplear un modelo de transición de estados, como es el modelo de Markov. Esta elección se debe a la extensión del horizonte temporal y naturaleza de la enfermedad, que implica que los pacientes puedan experimentar fases de estabilidad (preprogresión con o sin tratamiento) y fases de progresión (progresión). Esta elección está en línea con las recomendaciones elaboradas por la Sociedad Internacional de Farmacoeconomía e Investigación de Resultados, en su Task Force, en el cual se indica que “si la conceptualización del problema de salud implica representar la enfermedad o el proceso de tratamiento como una serie de estados de salud, los modelos de transición de estado son apropiados” [11]. Asimismo, se indica que los modelos de transición son particularmente útiles en el caso de horizontes temporales amplios, como es el caso en el presente análisis11. Finalmente, un reciente estudio sugiere que, en comparación con otros tipos de diseños, los modelos de Markov resultan más adecuados cuando el análisis requiere la simulación de varias líneas de tratamiento consecutivas [12].
Este estudio no está exento de limitaciones. Debido a la ausencia de ensayos clínicos controlados que evalúen la eficacia del retratamiento con bortezomib en pacientes con mieloma múltiple previamente tratados, los datos relativos a la eficacia de bortezomib en combinación con dexametasona han sido extraídos de un estudio retrospectivo de retratamiento con bortezomib, realizado en pacientes con mieloma múltiple de Alemania y Suiza [15]. El tamaño muestral (n = 42) y el diseño del estudio pueden introducir cierta incertidumbre en el análisis realizado. No obstante, el estudio aporta datos relativos a la efectividad de bortezomib en la práctica clínica habitual, lo que confiere una mayor validez externa al análisis y generalización de los resultados. Asimismo, la concordancia de los resultados de este estudio con los realizados en el entorno noruego y griego indica que la elección del estudio retrospectivo de retratamiento con bortezomib, para la estimación de la eficacia de bortezomib no comporta una subestimación de los beneficios clínicos asociados a este tratamiento. Otra posible limitación de este análisis se relaciona con las características de la población incluida en los ensayos sobre mieloma múltiple MM-009 y MM-010, de los que se han extraído los datos de eficacia relativos al tratamiento con lenalidomida en combinación con dexametasona [13],[14]. En los estudios mencionados, una menor proporción de pacientes había recibido una única terapia previa antes de recibir el tratamiento con lenalidomida en combinación con dexametasona, por lo que la población incluida en los ensayos podría diferir de la población de interés. Sin embargo, el NICE Evidence Review Group, tras la revisión del presente estudio en su adaptación al Reino Unido, determinó que a pesar de estas diferencias, los resultados de los estudios sobre mieloma múltiple MM-009 y MM-010 podían ser extrapolados a la población de interés sin comprometer la generalización de los resultados [9].
En conjunto, pese a la posible incertidumbre asociada a los datos de eficacia a partir de los cuales se ha realizado la comparación entre los tratamientos evaluados, los análisis de sensibilidad indican que los resultados obtenidos son robustos. En concreto, el análisis de sensibilidad univariante muestra como en la mayoría de las variables evaluadas, la variación introducida en los valores del caso base, no afecta al costo-efectividad de lenalidomida en combinación con dexametasona frente a bortezomib. Asimismo, de acuerdo a los resultados del análisis de sensibilidad probabilístico, que evalúa el impacto de la variación simultánea de todos los parámetros del modelo en los resultados del análisis, el tratamiento con lenalidomida en combinación con dexametasona presenta un 98,4% de probabilidades de ser costo-efectivo frente a bortezomib, teniendo en cuenta una disposición a pagar de 27 410 507,70 pesos chilenos (45 258 dólares).
Conclusión
Este estudio sugiere que lenalidomida en combinación con dexametasona representa una alternativa potencialmente eficiente en el tratamiento en segunda línea de pacientes con mieloma múltiple no candidatos a trasplante desde la perspectiva del sistema público nacional de salud chileno, al proporcionar una mayor sobrevida y al suponer un costo por año de vida ganado inferior a un PIB per cápita y un costo por año de vida ganado ajustado por calidad inferior a tres veces el PIB per cápita chileno.
Notas
Roles de autoría
MA: Recopilación de datos, Metodología, Validación, Visualización, Redacción de borrador original, Redacción-revisión y edición. IG: Recopilación de datos, Metodología, Validación, Redacción de borrador original, Redacción-revisión y edición. LL: Metodología, Administración de proyectos, Supervisión, Validación, Redacción-revisión y edición. SA: Metodología, Administración de proyectos, Supervisión, Validación, Redacción-revisión y edición.
Agradecimientos
Se agradecen a la Dra. Camila Peña y Dra. Carolina Guerra por su participación en el proyecto, validando y aportando nuevos datos clínicos y económicos, que han permitido la adaptación del presente trabajo al ámbito sanitario público chileno.
Financiamiento
El presente trabajo de investigación fue financiado por la empresa Tecnofarma S.A. La empresa no tuvo influencia en el diseño del estudio, análisis o interpretación de los datos. Asimismo, no hubo influencia en la preparación, revisión o aprobación del manuscrito.
Declaración de conflicto de intereses
Los autores han completado el formulario de declaración de conflictos de intereses del ICMJE, y declaran haber recibido financiamiento de parte de Tecnofarma S.A para la realización del estudio de evaluación económica. Los formularios pueden ser solicitados contactando al autor responsable o a la dirección editorial de la Revista.

