Análisis
← vista completaPublicado el 11 de julio de 2019 | http://doi.org/10.5867/medwave.2019.06.7664
La revisión sistemática como herramienta para la validación científica de propiedades saludables de los alimentos y factores alimentarios
Using systematic reviews in the scientific substantiation of health properties of foods and food constituents
Resumen
Las revisiones sistemáticas de literatura constituyen una de las principales metodologías utilizadas en la validación de las propiedades saludables de los alimentos, o factores alimentarios, que afectan la fisiología humana. Esta herramienta, basada en la evidencia obtenida a través de ensayos clínicos aleatorizados realizados con un diseño experimental adecuado, permite concluir si existe una relación causal entre el producto consumido y un efecto beneficioso para la salud, principio que sustenta el calificativo de los alimentos como “funcionales”. Se presentan y analizan las características y la forma en la que las revisiones sistemáticas pueden contribuir a que las agencias regulatorias aprueben un mensaje saludable (health claim), dirigido al consumidor.
|
Ideas clave
|
Introducción
Los alimentos no solamente son vehículos de nutrientes y energía, sino que además proporcionan placer y bienestar; y, lo que es muy importante, aportan componentes que ejercen acciones fisiológicas en el organismo que van más allá de su aporte nutricional. Estas propiedades, denominadas saludables o funcionales, otorgadas por compuestos químicos “bioactivos” contenidos en una matriz alimentaria, constituyen un tema que ha ocupado la atención de los investigadores, los consumidores, la industria agroalimentaria y los legisladores durante muchos años. En este contexto, es especialmente relevante la promoción de los beneficios para la salud mediante mensajes dirigidos al consumidor, utilizando diversas técnicas de marketing. Los alimentos denominados funcionales, poseen atributos saludables que deben ser comprobados científicamente. Sin embargo, muchas veces no se cuenta con una evidencia lo suficientemente robusta que sustente las declaraciones o mensajes. Esta situación ha llevado a que las entidades regulatorias de países o grupos de países, establezcan criterios claros al respecto[1],[2].
Para hacer una declaración (mensaje saludable o health claim) que asocie el consumo de un alimento o factor alimentario con un efecto beneficioso, en relación con alguna enfermedad o condición de salud, primero debe demostrarse esta acción. Solo de esta forma se podrá lograr que las agencias regulatorias, tales como la EFSA (Agencia de Inocuidad Alimentaria Europea) (http://www.efsa.europa.eu/), la FDA (Agencia de Drogas y Alimentos de EEUU) (https://www.fda.gov/) o el Ministerio de Salud, Trabajo y Bienestar de Japón (https://www.mhlw.go.jp/english/), aprueben un mensaje saludable. En Chile, el organismo encargado de aprobar el uso de mensajes saludables es el Ministerio de Salud (https://www.minsal.cl/), de acuerdo con el Reglamento Sanitario de Alimentos (Decreto 977/96 y sus modificaciones).
Por su parte, el Codex Alimentarius (http://www.fao.org/fao-who-codexalimentarius/es/), referente en normativa alimentaria a nivel mundial, define la declaración de propiedades saludables como “cualquier representación que declara, sugiere o implica que existe una relación entre un alimento, o un constituyente de dicho alimento, y la salud”. Añade que “las declaraciones de propiedades saludables y funcionales deben estar apoyadas por una cantidad de evidencia científica válida y suficiente como para justificarlas, proveer información verídica y no engañosa para ayudar al consumidor a elegir dietas saludables, y ser apoyadas por una educación específica para el consumidor”. No obstante, hoy en día existe una gran promoción de alimentos y factores alimentarios a los que se les atribuyen propiedades beneficiosas para la salud, sin contar con la adecuada validación científica de estos efectos.
Las revisiones sistemáticas consisten en la búsqueda y compilación de evidencia empírica con criterios preestablecidos para responder una determinada pregunta de interés[3], permitiendo apoyar la nutrición basada en evidencia[4]. Por lo tanto, el presente artículo tiene por objetivo describir la metodología de las revisiones sistemáticas de la literatura y su rol en la validación de los posibles efectos saludables de estos productos, ya sea que se hayan extraído y aislado de su fuente original o que se encuentren incluidos en una matriz alimentaria, que es la forma habitual de consumo, formando parte de una dieta habitual.
Aportes de los distintos diseños metodológicos cuantitativos a la investigación en alimentos funcionales
La epidemiología clínica ha provisto una sistematización de distintos diseños metodológicos que dan cuenta de preguntas de investigación. Los estudios observacionales, como los de corte transversal, de casos y controles y de cohorte, aportan información relevante. Sin embargo, no permiten determinar causalidad y están sujetos a una gran cantidad de sesgos que pueden afectar los resultados obtenidos[5],[6]. En consecuencia, en el caso del estudio de los efectos fisiológicos de los alimentos, los diseños observacionales no constituyen la metodología más apropiada para este propósito.
Si lo que se busca es analizar una intervención dietética sobre una condición fisiológica específica, el diseño a elegir será un ensayo clínico aleatorizado (RCT por sus siglas en inglés, Randomized Clinical Trial), nombre que reciben los estudios experimentales en epidemiología clínica[7],[8]. En ellos, el factor de exposición (alimento o factor alimentario) es asignado aleatoriamente a un grupo de personas, mientras que un grupo de comparación recibe un producto semejante que no contiene el factor alimentario (placebo o comparador). La aleatorización constituye un hecho central en el control de los factores de confusión, puesto que pretende homogeneizar las condiciones basales de los participantes en el estudio, lo que reduce la probabilidad de que la asociación observada sea explicada por una variable distinta a la intervención[9]. Adicionalmente, su carácter prospectivo cumple con el criterio de temporalidad, dado que se sabe a ciencia cierta que la exposición precede al efecto, lo que daría respuesta a las hipótesis de causalidad[8]. A los ensayos clínicos aleatorizados se les ha llamado “la piedra angular” de la salud basada en evidencia, pues son el componente central de las revisiones sistemáticas dedicadas a responder preguntas de investigación asociadas a intervenciones. Debido a que las revisiones sistemáticas integran los resultados obtenidos por múltiples ensayos clínicos aleatorizados, refinan la estimación del tamaño del efecto, aportando un nivel mayor de evidencia científica para la evaluación de mensajes saludables al compararse con un único ensayo clínico aleatorizado. La pirámide de la jerarquía de la calidad de la evidencia científica en ciencias de la salud puede observarse en la Figura 1[10].
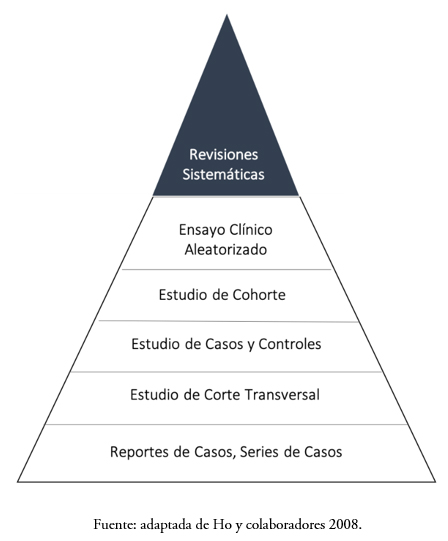 Tamaño completo
Tamaño completo De asociación a causalidad: del modelo preclínico al organismo humano
En 1965, el epidemiólogo británico Austin Bradford Hill enunció el “principio de coherencia”, en el contexto de la presentación de nueve aspectos de asociación que responderían a su pregunta “¿en qué circunstancias podemos pasar de una asociación observada a un veredicto de causalidad?”[11]. Estos aspectos, reconocidos posteriormente como los criterios de Bradford Hill[12], dan cuenta de una mayor probabilidad de que la asociación observada entre dos factores sea causal. El principio de coherencia manifiesta que una conclusión causal no debería contradecir en sus fundamentos al conocimiento del momento[12]. Si bien este concepto es materia de discusión y debate, puede entenderse como la necesidad de que exista una congruencia entre la evidencia aportada por los estudios preclínicos (in vitro, in vivo) y los estudios clínicos. De no existir este correlato, se pondría en entredicho el establecimiento de una relación de causalidad.
Un buen ejemplo está dado por el caso de los compuestos fenólicos contenidos en alimentos. Si bien se han demostrado múltiples efectos potencialmente beneficiosos de diversas moléculas de este tipo en ensayos in vitro y en modelos animales, atribuidos a sus propiedades antioxidantes, antiinflamatorias, anticancerígenas, cardioprotectoras, neuroprotectoras, entre otras, esto no ha sido sistemáticamente observado a nivel clínico[13]. A la fecha, los estudios en seres humanos solo han logrado validar científicamente dos mensajes saludables para polifenoles que han sido aprobados por las agencias regulatorias, que corresponden al hidroxitirosol del aceite de oliva[14] y los flavanoles del cacao[15]. Así, la falta de coherencia entre los ensayos de laboratorio y los estudios en seres humanos, imposibilita la validación científica de los efectos fisiológicos que ejercen estos compuestos[16],[17].
Actualmente, para considerar como verdadera a una inferencia causal, la investigación debe integrar hallazgos de múltiples disciplinas científicas[18]. En este sentido, es fundamental contar con estudios en humanos con diseños metodológicos que analicen la efectividad de una intervención y que tengan la capacidad de establecer nexos de causalidad; que, como ha sido referido, está dada principalmente por los ensayos clínicos aleatorizados y con mayor robustez por las revisiones sistemáticas de ensayos clínicos aleatorizados. A modo de ejemplo, el Ministerio de Salud, Trabajo y Bienestar de Japón, establece específicamente que para validar la efectividad de un producto puede recurrirse a dos métodos: ensayos clínicos aleatorizados o revisiones sistemáticas de ensayos clínicos aleatorizados acerca del producto terminado o los componentes que ejercen el efecto (http://www.caa.go.jp/foods/index23.html).
Revisiones sistemáticas de la literatura
Las revisiones sistemáticas constituyen un método ampliamente usado en la investigación biomédica aplicada a los efectos farmacológicos, entre otras intervenciones terapéuticas o dirigidas a mejorar la salud de las personas. Asimismo, esta herramienta es muy útil para evaluar la evidencia científica en relación con los efectos fisiológicos que ejerce el consumo de componentes bioactivos alimentarios en el organismo humano. Las revisiones sistemáticas permiten responder si se produce un efecto fisiológico o clínico específico mediante una metodología estandarizada y reproducible, que da cuenta de un protocolo de investigación fijado a priori[19]. Éstas incluyen varias etapas[20],[21],[22],[23],[24]: en un principio, se plantea una hipótesis con objetivos claros alrededor de una pregunta de investigación a responder. Se desarrolla una búsqueda exhaustiva de trabajos primarios (ensayos clínicos aleatorizados) en bases de datos pertinentes y también en literatura no publicada (“literatura gris”), tales como resúmenes de congresos u otros documentos[25]. Se identifican los ensayos clínicos aleatorizados que responden a la pregunta formulada, con una selección de acuerdo a criterios preestablecidos[26], por ejemplo, los trabajos incluidos debieran tener consistencia en el producto evaluado con respecto a la manera habitual y forma de su consumo, y la matriz alimentaria. Además de incluir trabajos de una intervención bien definida, se debe contemplar una población semejante, bien caracterizada, junto con desenlaces de interés bien definidos (biomarcador de efecto o un efecto clínico)[27],[28]. Etapas posteriores incluyen evaluación de la calidad metodológica y el análisis del riesgo de sesgo de cada estudio por medio de instrumentos estandarizados[29], extracción de datos desde los estudios primarios, síntesis de la evidencia recogida e interpretación de los hallazgos.
Es posible realizar un análisis estadístico (cuantitativo), el denominado metanálisis, donde se sintetizan y comparan los principales estimadores de cada estudio considerado en la revisión, de modo de generar un estimador combinado que integre los resultados de los estudios, siempre que su heterogeneidad lo permita[30]. En la etapa de conclusiones, la aplicación de la metodología GRADE (Grades of Recommendation, Assessment, Development and Evaluation) permite la elaboración de tablas de resumen de hallazgos (Summary of Findings) (http://www.gradeworkinggroup.org/), estableciendo una clasificación que hace referencia a la certeza de la totalidad de la evidencia, a partir de la evaluación de la calidad de los trabajos incluidos. En este esquema, la puntuación de la calidad de la evidencia disminuye si el cuerpo de la evidencia contiene ensayos clínicos aleatorizados de baja calidad, y eventualmente se podrían incluir estudios observacionales que hayan controlado los sesgos importantes durante su ejecución.
Murad y colaboradores[31] han propuesto una nueva conceptualización de la jerarquía de la evidencia tradicional (Figura 1), reconociendo el rol de las revisiones sistemáticas en la integración de la evidencia existente con el propósito de llevar los resultados a su aplicación, como un lupa a través de la cual se evalúa la evidencia (Figura 2). Además, este esquema hace referencia a la calificación mediante la metodología GRADE, pues presenta líneas onduladas entre los diseños metodológicos en la jerarquía, reflejando que la calidad de la evidencia es fluctuante de acuerdo con los estudios incluidos.
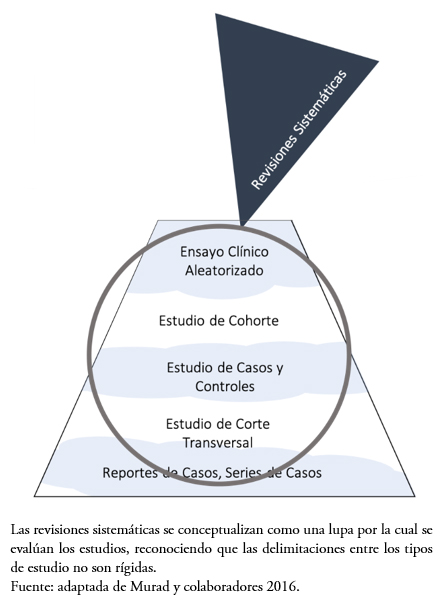 Tamaño completo
Tamaño completo Consideraciones finales
Las revisiones sistemáticas son una herramienta de gran utilidad en la validación de efectos fisiológicos de alimentos o factores alimentarios, permitiendo sustentar científicamente sus propiedades funcionales. Se basan en los resultados de ensayos clínicos aleatorizados que cumplen con requisitos rigurosos en su diseño experimental, y permiten realizar una asociación causal entre el alimento y el efecto fisiológico evaluado.
Si los alimentos o algunos de sus componentes solo demuestran efectos en ensayos de laboratorio, pruebas in vitro o en modelos animales, como es el caso de numerosos compuestos bioactivos alimentarios (polifenoles, carotenoides, compuestos azufrados, entre otros), sin coherencia con resultados de los ensayos clínicos aleatorizados, no es posible validar su efecto y no cabría denominarlos alimentos funcionales. Esta falta de coherencia se refleja en el escaso número de mensajes saludables aprobados por las agencias regulatorias a nivel internacional, en contraste con una gran demanda.
El reconocimiento de las revisiones sistemáticas en la validación científica de propiedades saludables en el ámbito de los alimentos está siendo reconocido cada vez más por los interesados en este tema, no solo a nivel de la academia, sino también por la industria, las agencias regulatorias y los consumidores. La aplicación de metodologías como las revisiones sistemáticas permite entregar información relevante a los tomadores de decisiones. Además, son una herramienta fundamental en la comunicación veraz de las propiedades de los alimentos o factores alimentarios a los consumidores, previniendo la confusión, la desinformación y el engaño.
Notas
Roles y contribuciones de los autores
ML: conceptualización, metodología, investigación, escritura (preparación del borrador original), escritura (revisión y edición), visualización, supervisión, administración del proyecto.
MA: conceptualización, metodología, investigación, escritura (preparación del borrador original), escritura (revisión y edición), visualización, supervisión, administración del proyecto.
JS: conceptualización, metodología, investigación, escritura (preparación del borrador original), escritura (revisión y edición), visualización, supervisión, administración del proyecto.
Financiamiento
Los autores declaran que no hubo fuentes externas de financiamiento.
Conflictos de intereses
Los autores completaron la declaración de conflictos de interés de ICMJE y declararon que no recibieron fondos por la realización de este artículo; no tienen relaciones financieras con organizaciones que puedan tener interés en el artículo publicado en los últimos tres años y no tienen otras relaciones o actividades que puedan influenciar en la publicación del artículo. Los formularios se pueden solicitar contactando al autor responsable o al Comité Editorial de la Revista.

