Resúmenes Epistemonikos
← vista completaPublicado el 29 de septiembre de 2016 | http://doi.org/10.5867/medwave.2016.6555
¿Es efectiva la N-acetilcisteína en el tratamiento de la fibrosis pulmonar?
Is N-acetylcysteine effective in the treatment of pulmonary fibrosis?
Resumen
La fibrosis pulmonar idiopática es una enfermedad respiratoria crónica y progresiva, que en etapas finales conlleva una alta mortalidad. Se han planteado múltiples opciones terapéuticas, entre ellas la N-acetilcisteína, pero su rol no está claramente establecido. Utilizando la base de datos Epistemonikos, la cual es mantenida mediante búsquedas en 30 bases de datos, identificamos ocho revisiones sistemáticas que en conjunto incluyen 16 estudios aleatorizados. Realizamos un metanálisis y tablas de resumen de los resultados utilizando el método GRADE. Concluimos que N-acetilcisteína probablemente aumenta el riesgo de hospitalización y exacerbaciones. Si bien no está claro si esto conlleva un aumento de mortalidad porque la certeza de la evidencia es muy baja, en general existe consenso en que no debiera utilizarse, a no ser que sea en el contexto de un nuevo estudio clínico.
Problema
En el último tiempo ha surgido gran cantidad de evidencia en torno a la efectividad de distintas terapias para la fibrosis pulmonar idiopática, sobre todo considerando que ninguna intervención más que el trasplante pulmonar aumenta claramente la sobrevida [1].
La N-acetilcisteína, por su carácter reductor, ejerce una actividad citoprotectora en el aparato respiratorio humano, actuando frente a la acción perjudicial del estrés oxidativo generado por radicales libres de diversa etiología. En base a su estructura derivada de la cisteína, la N-acetilcisteína tiene un rol precursor en la síntesis de la molécula antioxidante glutatión y normaliza sus niveles cuando se ven reducidos por una acción oxidante continua sobre el aparato respiratorio. Es así como en los pacientes con fibrosis pulmonar idiopática la acción de este fármaco podría estar mediada por el mecanismo de acción previamente descrito [2].
Además, N-acetilcisteína está disponible ampliamente, y según la guía ATS/ERS 2011, usada sola o en combinación (con prednisolona y azatriopina) podría ser una elección razonable en una minoría de pacientes con esta enfermedad [1]. Sin embargo, una actualización de la misma guía el año 2015 propone prohibir su uso debido a un aumento del riesgo de hospitalización y muerte [3].
Métodos
Utilizamos la base de datos Epistemonikos, la cual es mantenida mediante búsquedas en 30 bases de datos, para identificar revisiones sistemáticas y sus estudios primarios incluidos. Con esta información generamos un resumen estructurado, siguiendo un formato preestablecido, que incluye mensajes clave, un resumen del conjunto de evidencia (presentado como matriz de evidencia en Epistemonikos), metanálisis del total de los estudios, tablas de resumen de resultados con el método GRADE, y tablas de otras consideraciones para la toma de decisión.
|
Mensajes clave
|
Acerca del conjunto de evidencia para esta pregunta
|
Cuál es la evidencia. |
Encontramos ocho revisiones sistemáticas publicadas en nueve referencias [4],[5],[6],[7],[8],[9],[10],[11],[12] que incluyen 16 estudios controlados aleatorizados publicados en 18 referencias [13],[14],[15],[16],[17],[18],[19],[20], [21],[22],[23],[24],[25],[26],[27],[28],[29],[30]. |
|
Qué tipo de pacientes incluyeron los estudios |
Todos los estudios incluyeron pacientes mayores de 18 años. En trece estudios el diagnóstico se realizó solo en base a las normas ATS/ERS/JRS/ALAT [13],[15],[20],[21],[22], [23],[24],[25],[26],[27],[28],[29],[30], en un estudio se incluyeron además pacientes con diagnóstico por imágenes o biopsia [16], en otro estudio se requirió además marcadores de injuria pulmonar [14] y en otro estudio el diagnóstico se realizó solo en base a imágenes y biopsia [18]. En tres estudios se incluyeron pacientes según test de función pulmonar o gases en sangre arterial [14],[16], [18], y un estudio especificó un score de disnea como criterio de inclusión [18]. |
|
Qué tipo de intervenciones incluyeron los estudios |
Dos estudios utilizaron N-acetilcisteína como monoterapia oral [15],[17], un estudio la utilizó como monoterapia inhalatoria [14], diez estudios la utilizaron en combinación con prednisona 0,4 a 0,5 mg/kg/día [13],[20],[21],[23], [24],[25],[26],[27],[30], un estudio la utilizó asociada con interferón [22], un estudio refiere asociar N-acetilcisteína a antiinflamatorios pero no especifica cuales [29], un estudio la utilizó asociada con prednisolona y azatriopina [18], y un estudio utilizó N-acetilcisteína asociada con prednisolona y azatriopina, el cual fue interrumpido precozmente por alta tasa de mortalidad [16]. Todos los estudios compararon contra placebo o tratamiento estándar (placebo, prednisolona y azatriopina), excepto un estudio que comparó con bromhexina [15]. |
|
Qué tipo de desenlaces midieron |
Las distintas revisiones sistemáticas identificadas agruparon los desenlaces de la siguiente forma:
|
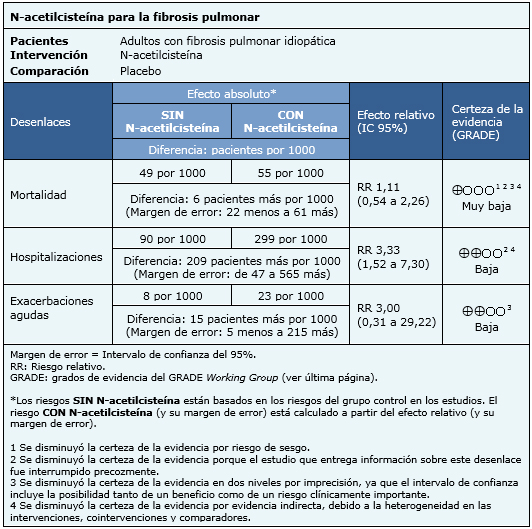
Resumen de los resultados
La información sobre los efectos de N-acetilcisteína está basada en cuatro estudios aleatorizados [14],[15],[16],[18] que incluyen 694 pacientes. El resto de los estudios no presentaba datos sobre los desenlaces de interés, o no fue posible utilizarlos en un metanálisis. Cuatro estudios [14],[15],[16],[18] midieron el desenlace mortalidad, y sólo un estudio [16] midió el desenlace hospitalizaciones y exacerbaciones agudas. El resumen de los resultados es el siguiente:
- No está claro si el uso de N-acetilcisteína aumenta o disminuye la mortalidad porque la certeza de la evidencia es muy baja.
- N-acetilcisteína podría aumentar el riesgo de hospitalizaciones, sin embargo, la certeza de la evidencia es baja..
- N-acetilcisteína podría aumentar el riesgo de exacerbaciones agudas, sin embargo, la certeza de la evidencia es baja..

Otras consideraciones para la toma de decisión
|
A quién se aplica y a quién no se aplica esta evidencia |
|
| Sobre los desenlaces incluidos en este resumen |
|
| Balance riesgo/beneficio y certeza de la evidencia |
|
| Qué piensan los pacientes y sus tratantes |
|
| Consideraciones de recursos |
|
| Diferencias entre este resumen y otras fuentes |
|
| ¿Puede que cambie esta información en el futuro? |
|
Cómo realizamos este resumen
Mediante métodos automatizados y colaborativos recopilamos toda la evidencia relevante para la pregunta de interés y la presentamos en una matriz de evidencia.
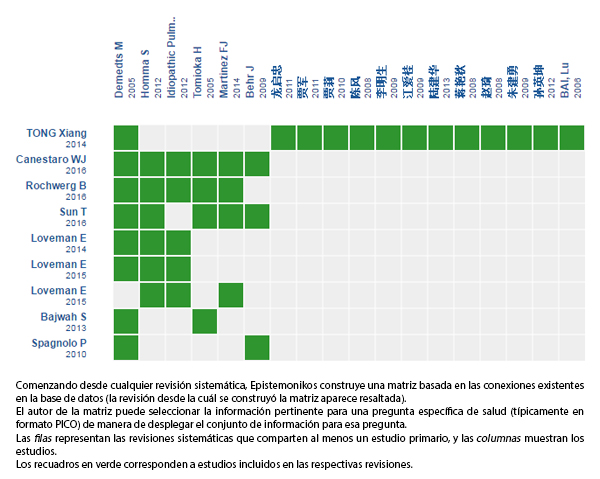
Siga el enlace para acceder a la versión interactiva: N-acetilcisteína para la fibrosis pulmonar idiopática
Notas
Si con posterioridad a la publicación de este resumen se publican nuevas revisiones sistemáticas sobre este tema, en la parte superior de la matriz se mostrará un aviso de “nueva evidencia”. Si bien el proyecto contempla la actualización periódica de estos resúmenes, los usuarios están invitados a comentar en Medwave o contactar a los autores mediante correo electrónico si creen que hay evidencia que motive una actualización más rápida.
Luego de crear una cuenta en Epistemonikos, al guardar las matrices recibirá notificaciones automáticas cada vez que exista nueva evidencia que potencialmente responda a esta pregunta. El detalle de los métodos para elaborar este resumen están descritos aquí: http://dx.doi.org/10.5867/medwave.2014.06.5997.
La Fundación Epistemonikos es una organización que busca acercar la información a quienes toman decisiones en salud, mediante el uso de tecnologías. Su principal desarrollo es la base de datos Epistemonikos (www.epistemonikos.org).
Los resúmenes de evidencia siguen un riguroso proceso de revisión por pares interno.
Declaración de conflictos de intereses
Los autores declaran no tener conflictos de intereses con la materia de este artículo.

