Suplementos
← vista completaPublicado el 21 de diciembre de 2016 | http://doi.org/10.5867/medwave.2016.6810
Factores de riesgo de fibrilación auricular en el post operatorio de cirugía de revascularización coronaria: papel de la apnea obstructiva del sueño
Risk factors for post coronary artery bypass graft atrial fibrillation: role of obstructive sleep apnea
Resumen
En este capítulo comenzamos analizando la cirugía de revascularización miocárdica y su complicación más frecuente, la fibrilación atrial postoperatoria. Analizamos los principales factores de riesgo para la fibrilación atrial postoperatoria y luego discutimos detalladamente la apnea obstructiva del sueño como factor riesgo. En este empeño, describimos cómo se diagnostica la apnea obstructiva del sueño, su pato-fisiología en relación con la fibrilación atrial postoperatoria y los estudios clínicos que recientemente han investigado la asociación entre la apnea obstructiva del sueño y fibrilación atrial postoperatoria. Concluimos con las estrategias de prevención y tratamiento de la fibrilación atrial postoperatoria y la discusión de futuras recomendaciones investigativas.
Introducción
La cirugía de revascularización coronaria (cirugía de revascularización coronaria) es uno de los procedimientos quirúrgicos más comunes, solamente en Estados Unidos se realizan más de 50 millones de cirugía de revascularización coronaria anualmente [1]. La fibrilación auricular post cirugía de revascularización coronaria sigue siendo la complicación más común después en este tipo de cirugía, estimándose que afecta entre un 20-50% de los pacientes [2],[3],[4],[5],[6],[7],[8]. La fibrilación auricular es una taquiarritmia supraventricular caracterizada por la activación auricular desordenada, que usualmente evoluciona con deterioro de la función auricular. La fibrilación atrial postoperatoria se asocia a una serie de complicaciones, como la duplicación del riesgo de mortalidad hospitalaria y a seis meses, triplica el riesgo de accidente cerebrovascular postoperatorio, duplica el riesgo de infarto de miocardio; así como un aumento de la morbilidad, las admisiones en las unidades de cuidados intensivos, la estancia en cuidados intensivos, la estancia hospitalaria, los costos del sistema de salud y la “carga” biopsicosocial para pacientes y familiares [2],[3],[4],[5],[6],[7],[8],[9].
Factores de riesgo
Varios estudios han investigado los factores que predicen la ocurrencia de fibrilación atrial postoperatoria. Nos centraremos en los factores predictivos más significativos, los cuales se pueden dividir en preoperatorios, intraoperatorios y postoperatorios. La apnea obstructiva de sueño se omitirá inicialmente, con la intención de analizarla detalladamente en las siguientes secciones.
Preoperatorio
La edad avanzada es el factor de riesgo independiente más fuertemente asociado con fibrilación atrial postoperatoria. Mathew y colaboradores demostraron que las probabilidades de desarrollar fibrilación atrial postoperatoria aumentan en un 75% por 10 años de incremento en la edad (intervalo de confianza del 95%: 59% al 93%), y que cualquier persona mayor de 70 años tiene alto riesgo de desarrollar fibrilación atrial postoperatoria [3]. El meta-análisis de Yin y colaboradores mostró que los pacientes con fibrilación atrial postoperatoria tienen un aumento de 5,04 puntos (intervalo de confianza del 95%: 4,16 a 5,91) en la diferencia de la edad media, pero el resultado fue heterogéneo [10]. Sin embargo, todos los estudios incluidos en el meta-análisis mostraron un aumento significativo en la edad de los pacientes con fibrilación atrial postoperatoria [10]. Este estudio también identificó a la hipertensión arterial y el infarto de miocardio preoperatorio como factores significativamente asociados a la ocurrencia de fibrilación atrial postoperatoria, pero en el caso de la hipertensión los resultados fueron muy heterogéneos.
Hernández y colaboradores realizaron un meta-análisis para investigar la obesidad como factor de riesgo de fibrilación atrial postoperatoria. Demostraron que los pacientes obesos tenían un 12% más de probabilidades de desarrollar esta complicación que los pacientes no obesos (IC del 95%: 4% a 21%), con una heterogeneidad moderada [11]. Hasta donde sabemos, no se han llevado a cabo meta-análisis sobre otros factores de riesgo preoperatorios. No obstante, algunos estudios han demostrado asociaciones significativas para muchas otras variables preoperatorias. Entre los que se incluyen el sexo masculino [12], la fibrilación auricular pre-operatoria [3],[12], otras arritmias [8], el crecimiento de la aurícula izquierda (AI) [13], la disfunción mecánica de la AI [14], la enfermedad pulmonar obstructiva crónica [2],[3],[15], la cardiopatía reumática [15], la insuficiencia cardíaca [5],[12],[16], la reducción de la fracción de eyección [4], y el uso preoperatorio de digoxina [2],[15].
Intraoperatoria
Varios estudios han identificado factores de riesgo intraoperatorios para el desarrollo de fibrilación atrial postoperatoria. Entre los que se incluyen tiempos prolongados de bomba [12],[15], ventilación [8] y circulación extracorpórea [8], así como la protección insuficiente de las aurículas [2] y el tiempo de circulación pulmonar [2],[12]. Existen evidencias contradictorias sobre el tipo de cardioplejía como factor de riesgo para el desarrollo de fibrilación atrial postoperatoria. Algunos estudios la reportan como un factor de riesgo [5], pero otros no le identifican una asociación significativa con el desarrollo de esta complicación [12]. Curiosamente, Almassi y colaboradores encontraron que la cardioplejia anterógrada caliente y la composición de la cardioplejia fueron factores de riesgo significativos, pero no la cardioplejia anterógrada fría, ni el método de cardioplejia [2].
Postoperatorio
Los factores postoperatorios identificados como predictores de fibrilación atrial postoperatoria incluyen la hipopotasemia [17], la hipomagnesemia [18], la estimulación eléctrica auricular postquirúrgica [12], la retirada de beta-bloqueadores [3], y la discontinuación de los inhibidores de la enzima convertidora de la angiotensina [3]. La suspensión de los beta-bloqueadores después de la cirugía de revascularización coronaria podría precipitar fibrilación atrial postoperatoria por varias razones. En primer lugar, el uso de beta-bloqueadores después de cirugía de revascularización coronaria es una medida profiláctica contra el desarrollo fibrilación atrial postoperatoria (ver debajo la sección de prevención de fibrilación atrial postoperatoria). Por lo tanto, es probable que su suspensión aumente el riesgo de fibrilación atrial postoperatoria debido a la pérdida de sus funciones protectoras. En segundo lugar, si los pacientes estaban usando beta-bloqueadores durante mucho tiempo antes de la cirugía, su discontinuación perioperatoria puede inducir taquicardia refleja; sobre todo después de someterse a un factor de gran estrés fisiológico como es la cirugía de revascularización coronaria. La combinación de ambos factores probablemente favorece el desarrollo de fibrilación auricular postoperatoria.
Apnea obstructiva del sueño como factor de riesgo para fibrilación auricular post-cirugía
La apnea obstructiva del sueño es una enfermedad que ha sido recientemente identificada como predictor de fibrilación atrial postoperatoria. Analizar esta relación es importante ya que puede facilitar la optimización del manejo de la fibrilación atrial postoperatoria. Vamos a empezar por examinar cómo se diagnostica la apnea obstructiva del sueño, luego la probable relación fisiopatológica entre apnea obstructiva del sueño y fibrilación atrial postoperatoria, y posteriormente discutiremos las evidencias sobre esta asociación.
La apnea obstructiva del sueño tiene una prevalencia de aproximadamente 3-7% en Norteamérica [19]. A pesar de este alto valor, varios estudios sugieren que la apnea obstructiva del sueño es vulgarmente no diagnosticada [20]. Es importante detectar y diagnosticar la apnea obstructiva del sueño para evitar complicaciones y lograr mejores resultados. El Cuestionario de Berlín es una herramienta utilizada para la detección de apnea obstructiva del sueño, mientras que la polisomnografía es el “gold” estándar para el diagnóstico. Ambos, el Cuestionario de Berlín y la polisomnografía serán analizados sepradamente.
El Cuestionario de Berlín ha sido validado contra la polisomnografía, demostrando una sensibilidad del 86%, una especificidad del 77% y un valor predictivo positivo del 89%. Se compone de 10 preguntas sobre los factores de riesgo para la apnea obstructiva del sueño, divididas en tres categorías: 1) el comportamiento de ronquidos y apneas (6 puntos), 2) el tiempo de vigilia sueño o fatiga (3 puntos); y 3) la presencia de obesidad o hipertensión arterial (2 puntos). Los pacientes solo deben marcar una respuesta por pregunta. Si dos o más categorías son positivas (categorías 1 y 2: + 2 puntos; categoría 3: +1 punto), los pacientes están en un alto riesgo de presentar apnea obstructiva del sueño. Al ser la polisomnografía un procedimiento caro y engorroso, muchos centros interpretan a los individuos con un alto riesgo de apnea obstructiva del sueño como un equivalente de tener un diagnóstico efectivo de la enfermedad. Sin embargo, es importante señalar que el Cuestionario de Berlín no da un diagnóstico definitivo, lo cual sólo puede hacerse con polisomnografía [21].
La polisomnografía se utiliza como una herramienta diagnóstica en la medicina del sueño. Registra los cambios biofisiológicos durante el sueño, incluyendo el flujo de aire respiratorio, el esfuerzo respiratorio, y la oximetría de pulso. El indicador de severidad más comúnmente utilizado para la apnea obstructiva del sueño es el índice de apnea-hipopnea, una medida que cuantifica el número de episodios de apnea o hipopnea por hora de sueño. La apnea obstructiva del sueño se clasifica en leve, moderada y severa si el índice de apnea-hipopnea es de 5-15, 16-30 y > 30, respectivamente [22].
Fisiopatología
El mecanismo exacto que subyace en la fisiopatología de la fibrilación atrial postoperatoria está todavía bajo investigación. Sin embargo, la relación fisiopatológica entre apnea obstructiva del sueño y la fibrilación auricular se ha explorado de forma adecuada. La apnea obstructiva del sueño se caracteriza por la oclusión repetitiva de la vía aérea superior durante el sueño, predispone a los individuos al desarrollo de enfermedad cardiovascular mediante hipoxemia intermitente, inflamación, disfunción autonómica, y cambios en la presión intratorácica [23].
Hipoxemia
El mecanismo detrás del cual la hipoxemia provoca fibrilación auricular no es absolutamente comprendido, pero se cree que es debido a una lesión endotelial vascular continua a través de estrés oxidativo. Como resultado, los pacientes con apnea obstructiva del sueño presentan un estado de inflamación crónica, predisponiendo a la hipertensión arterial y fibrosis auricular [23].
Inflamación
Los pacientes con apnea obstructiva del sueño presentan tanto inflamación local, como sistémica. Se postula que la inflamación local del tracto respiratorio superior puede facilitar el estrechamiento de las vías respiratorias y la disfunción muscular, empeorando así la frecuencia y la intensidad de los episodios de apnea e hipoapnea. La inflamación sistémica puede potenciar las enfermedades cardiovasculares a través de niveles elevados de mediadores proinflamatorios [23].
Cambios en la presión intratorácica
El estrechamiento repetitivo de las vías respiratorias superiores puede generar grandes gradientes de presión, lo cual es causa de fibrosis auricular y dilatación de la aurícula - ambos predictores de fibrilación auricular. Los modelos animales de apnea obstructiva del sueño han demostrado que causa distensión de la aurícula izquierda, sustentando la idea de que los cambios de presión intratorácica pueden actuar como un sustrato para la fibrilación auricular [23].
Inestabilidad autonómica
La inestabilidad o disfunción autonómica es un reconocido mecanismo responsable de iniciar eventos cardiovasculares incluyendo fibrilación auricular, otras arritmias, insuficiencia cardíaca, infarto de miocardio, accidente cerebrovascular e incluso la muerte [23].
Además de actuar potencialmente como sustratos independientes de fibrilación auricular, los mecanismos antes mencionados pueden causar remodelado auricular, que es un factor de riesgo de perpetuación de la fibrilación auricular [23].
Estudios clínicos
Hasta hace poco, la literatura investigativa sobre la asociación entre apnea obstructiva del sueño y fibrilación atrial postoperatoria era escasa. Mooe y colaboradores [1996] fueron el primer grupo que estudió esta relación, encontrando que la apnea obstructiva del sueño es un predictor independiente de fibrilación atrial postoperatoria tras el análisis multivariado ajustado (odds ratio [OR] = 2,8; IC del 95%: 1,2 a 6,8) [24]. Otros estudios realizados posteriormente, lo confirmaron pero con menor potencia estadística. Van Oosten y colaboradores [2014] abordaron este tópico y demostraron que apnea obstructiva del sueño es un predictor independiente de fibrilación atrial postoperatoria. El grupo de Queen’s University utilizó el Cuestionario de Berlín para clasificar a los pacientes en alto y bajo riesgo de apnea obstructiva del sueño, y llevó a cabo estudios de polisomnografía en un subgrupo de la muestra (pacientes con apnea obstructiva del sueño confirmada). El 29,7% de los pacientes en el grupo de bajo riesgo desarrolló fibrilación atrial postoperatoria, mientras que las proporciones en los grupos de alto riesgo, alto riesgo + apnea obstructiva del sueño confirmada, y apnea obstructiva del sueño confirmada fueron significativamente más altas - 43,3%, 45,5% y 51,4%, respectivamente-. Esto demostró que el riesgo de fibrilación atrial postoperatoria aumentaba a medida que el diagnóstico de la apnea obstructiva del sueño era más seguro (por ejemplo, una mayor proporción de pacientes con apnea obstructiva del sueño confirmada desarrollaron fibrilación atrial postoperatoria en comparación con los clasificados como de alto riesgo por el Cuestionario de Berlín). El odds ratio ajustado multivariable para el grupo apnea obstructiva del sueño confirmada + alto riesgo en comparación con el grupo de bajo riesgo fue de 2,18 (intervalo de confianza del 95%: 1,30 a 3,65). Esto sugiere que la apnea obstructiva del sueño es un fuerte predictor de fibrilación atrial postoperatoria [25].
Qaddoura y colaboradores (del mismo grupo) realizaron un meta-análisis para sintetizar la evidencia acerca de la asociación entre apnea obstructiva del sueño y fibrilación atrial postoperatoria después de cirugía de revascularización coronaria electiva. Se identificaron cinco estudios de cohorte prospectivos con un total de 642 pacientes, algunos utilizando cuestionarios de evaluación y otros usando polisomnografía para diagnosticar apnea obstructiva del sueño. Al combinar todos los estudios, encontraron que apnea obstructiva del sueño se asocia con un mayor riesgo de desarrollar fibrilación atrial postoperatoria (OR, 1,86; intervalo de confianza del 95%: 1,24 a 2,80), con una heterogeneidad moderada. Tres análisis de subgrupos también fueron realizados. La magnitud de la asociación aumentó cuando: se utilizó la polisomnografía para valorar apnea obstructiva del sueño (OR, 2,34; intervalo de confianza del 95%: 1,48 a 3,70), cuando la apnea obstructiva del sueño grave se incluyó a partir de los datos de un único estudio (OR, 2,59; intervalo de confianza del 95%: 1,63 a 4,11), y cuando se combinaron los análisis ajustados (OR 2,38; intervalo de confianza del 95%: 1,57 a 3,62), sin detectarse heterogeneidad en el análisis de los subgrupos. La Tabla 1 resume los resultados de este meta-análisis. Los datos sugieren que el riesgo de fibrilación atrial postoperatoria aumenta más con el diagnóstico definitivo de apnea obstructiva del sueño, y potencialmente con una mayor severidad de esta última. Esto también proporciona confianza en la validez de los resultados para las asociaciones no ajustadas, al ser similares a la asociación ajustada multivariable [26]. Lo cual es importante dado el mencionado riesgo multifactorial de la fibrilación atrial postoperatoria.
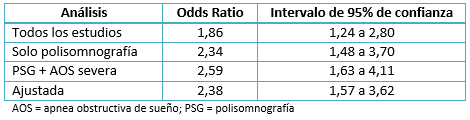 Tamaño completo
Tamaño completo Prevención y tratamiento de la fibrilación atrial postoperatoria
Varios estudios han investigado las formas de prevenir y tratar la fibrilación atrial postoperatoria. Al igual que en la sección sobre los factores de riesgo para fibrilación atrial postoperatoria, a continuación se analizarán sólo las principales estrategias preventivas y de tratamiento de la fibrilación atrial postoperatoria.
Prevención
Un meta-análisis de Arsenault y colaboradores demostró que los beta-bloqueadores logran la mayor magnitud preventiva de fibrilación atrial postoperatoria (OR, 0,33; intervalo de confianza del 95%: 0,26 a la 0,43), con una heterogeneidad moderada [27]. El Solatol (un beta-bloqueante con propiedades anti-arrítmicas Clase III según la clasificación de Vaughan Williams) tuvo una eficacia similar en la prevención de fibrilación atrial postoperatoria (OR, 0,34; intervalo de confianza del 95%: 0,26-0,43), con mucho menos heterogeneidad [27]. El magnesio presentó menos eficacia, con una OR de 0,55 (intervalo de confianza del 95%: 0,41 a 0,73), y heterogeneidad moderada [27]. Un meta-análisis de 20 ensayos controlados aleatorizados (ECA) realizados por Miller y colaboradores mostró valores similares para el magnesio (OR, 0,54; intervalo de confianza del 95%: 0,38 a 0,75), con heterogeneidad moderada [28]. La amiodarona (OR, 0,43; intervalo de confianza del 95%: 0,34 a 0,54), la estimulación eléctrica auricular (OR, 0,47; intervalo de confianza del 95%: 0,36 a 0,61) y la pericardiotomıa posterior (OR, 0,35; intervalo de confianza del 95%: 0,18 a 0,67) han sido reportados como efectivos, pero con relativamente alta heterogeneidad [27]. Un meta-análisis de Wijeysundera y colaboradores sobre los bloqueantes-cálcicos en la prevención de complicaciones cardiovasculares post-cirugía de revascularización coronaria, mostró que los no-dihidropiridinicos reducen significativamente las taquicardias supraventriculares en general (OR, 0,62; intervalo de confianza del 95%: 0,41 a 0,93), con una heterogeneidad significativa [29].
Otras medidas profilácticas reportadas por algunos ensayos incluyen las grasas poliinsaturadas y los agentes anti-inflamatorios. Un ensayo clínico aleatorizado realizado por Calò y colaboradores identificó que los ácidos grasos poliinsaturados N-3 protegen contra la fibrilación auricular post cirugía (OR, 0,32; intervalo de confianza del 95%: 0,10 a la 0,98) [30]. Cheruku y colaboradores realizaron un ensayo clínico aleatorizado pequeño para investigar los medicamentos anti-inflamatorios no esteroideos y encontraron un beneficio significativo con ibuprofeno, el 28,6% de los pacientes desarrolló fibrilación atrial postoperatoria en el grupo control versus el 9,8% en el grupo con ibuprofeno (p <0,017) [31]. En otro ensayo clínico aleatorizado Halonen y colaboradores hallaron que la hidrocortisona reduce significativamente la incidencia de fibrilación atrial postoperatoria (riesgo ajustado, 0,54; intervalo de confianza del 95%: 0,35 a 0,83) [32].
El meta-análisis de Arsenault y colaboradores también demostró que las intervenciones profilácticas disminuyen la estancia hospitalaria y el costo del tratamiento, y de manera no significativa el riesgo de accidente cerebrovascular postoperatorio [27]. Esto pone de relieve la importancia de adoptar medidas para prevenir el desarrollo de fibrilación atrial postoperatoria.
Tratamiento
El primer paso en el tratamiento de la fibrilación atrial postoperatoria, al igual que en muchos trastornos, es tratar las comorbilidades subyacentes. Esto incluye el tratamiento de las alteraciones electrolíticas, la enfermedad pulmonar obstructiva crónica, y la hipoxia [33]. El tratamiento posterior específico de la fibrilación atrial postoperatoria está indicado para pacientes que permanecen sintomáticos, hemodinámicamente inestables, desarrollan isquemia o insuficiencia cardiaca. Se discutirán cuatro estrategias convencionales de tratamiento: el control de la frecuencia ventricular, el control del ritmo, la cardioversión eléctrica, y la prevención del tromboembolismo.
Control de la frecuencia
La primera opción de tratamiento son los beta-bloqueantes de acción corta, especialmente en los pacientes con cardiopatía isquémica. Extrapolar las tradicionales contraindicaciones absolutas (pre-excitación ventricular, asma bronquial, hipotensión arterial y bloqueo aurículo-ventricular avanzado) y relativas (enfermedad pulmonar obstructiva crónica e insuficiencia cardíaca) de los beta-bloqueantes a los disimiles contextos que pueden existir en la fibrilación atrial postoperatoria puede ser un gran reto para equipo a cargo. Una alternativa es utilizar agentes bloqueantes del nodo aurículo-ventricular, como los bloqueantes-cálcicos no dihidropiridínicos. La digoxina se puede usar en pacientes con insuficiencia cardiaca, pero es menos eficaz cuando está elevado el tono adrenérgico. Por último, la amiodarona se asocia con el control efectivo de la frecuencia cardiaca, y mejoras del estado hemodinámico [33].
Control del ritmo
El control del ritmo tiene varias ventajas, incluyendo un menor tiempo para la cardioversión, la prolongación del mantenimiento del ritmo sinusal y la disminución de la duración de la estancia hospitalaria en general. Esta estrategia podría utilizar la cardioversión eléctrica en pacientes sintomáticos o cuando la respuesta ventricular es difícil de controlar. En la cardioversión farmacológica se emplean amiodarona, procainamida, ibutilida, y sotalol. El solatol ha demostrado ser eficaz en la reducción de la frecuencia ventricular, pero parece ser menos efectivo para inducir cardioversión de la fibrilación auricular [33].
Cardioversión eléctrica
Esta estrategia de tratamiento debe realizarse urgentemente si la fibrilación atrial postoperatoria provoca colapso hemodinámico, insuficiencia cardiaca aguda o isquemia miocárdica. Por otra parte, se puede utilizar de forma electiva cuando con la terapia farmacológica no se lograr recuperar el ritmo sinusal. El tromboembolismo es una preocupación importante en las cardioversiones farmacológica y eléctrica, por lo que se recomienda la anticoagulación durante 3 a 4 semanas antes de la cardioversión en población general si la fibrilación auricular dura más de 48 horas [33].
Prevención de eventos tromboembólicos
La fibrilación atrial postoperatoria se asocia con un mayor riesgo de accidente cerebrovascular perioperatorio, y esto potencialmente se puede reducir con anticoagulación. En contraste, la anticoagulación en el período post-operatorio puede aumentar el riesgo de sangrado o taponamiento cardiaco, y los médicos deben sopesar entre la duración a menudo transitoria y autolimitada de la fibrilación atrial postoperatoria y el riesgo de sangrado postoperatorio secundario a anticoagulación. El elevado riesgo de hemorragia puede superar el beneficio de la prevención del tromboembolismo en algunos pacientes, especialmente en aquellos con edad avanzada, hipertensión no controlada, e historia de sangrado [33].
La fibrilación atrial postoperatoria a menudo se resuelve espontáneamente en las primeras horas o en días. Por tanto, como regla general, la anticoagulación se inicia en episodios de fibrilación atrial postoperatoria prolongados (> 48 horas) y/o frecuentes [33]. La anticoagulación se recomienda especialmente para los pacientes con alto riesgo de tromboembolismo, incluyendo aquellos con fibrilación atrial postoperatoria y antecedentes de accidente cerebrovascular o ataque isquémico transitorio. El tratamiento anticoagulante en estos pacientes debe continuar durante 30 días después de recuperar el ritmo sinusal [34].
Recomendaciones futuras
La fibrilación atrial postoperatoria es un trastorno serio con muchas secuelas negativas. Sus factores de riesgo deben ser reconocidos para tratarla de manera óptima, incluyendo la detección y manejo de la apnea obstructiva del sueño. Varias estrategias han sido definidas para tratar y prevenir la fibrilación atrial postoperatoria, como se discutió en secciones anteriores. Una estrategia que aún no se ha explorado completamente es el uso de la presión positiva continua en vía aérea (CPAP) en la prevención de la fibrilación auricular post cirugía y sus complicaciones. La presión positiva continua en vía aérea se utiliza para el tratamiento de la apnea obstructiva del sueño, y su uso puede mejorar los resultados para los pacientes con apnea obstructiva del sueño y fibrilación atrial postoperatoria, sobre todo porque se identifica como un predictor significativo de fibrilación atrial postoperatoria. Por tanto, en el futuro debe investigarse el empleo de la presión positiva continua en vía aérea en el mejoramiento del pronóstico de los pacientes con fibrilación atrial postoperatoria.
A su vez, la evidencia hasta la fecha sugiere que la gravedad de la apnea obstructiva del sueño puede aumentar la magnitud de su asociación con fibrilación atrial postoperatoria. Los datos no son suficientes para definitivamente concluir que el riesgo de fibrilación atrial postoperatoria aumenta con la gravedad de la apnea obstructiva del sueño, lo cual estimula la realización de futuros estudios para explorar este importante tópico.
Notas
Declaración de conflictos de intereses
Los autores han completado el formulario de declaración de conflictos intereses del ICMJE traducido al castellano por Medwave, y declaran no haber recibido financiamiento para la realización del reporte; no tener relaciones financieras con organizaciones que podrían tener intereses en el artículo publicado, en los últimos tres años; y no tener otras relaciones o actividades que podrían influir sobre el artículo publicado. Los formularios pueden ser solicitados contactando al autor responsable o a la dirección editorial de la Revista.
Financiamiento
Los autores declaran que no hubo fuentes de financiación externas.

