Resúmenes Epistemonikos
← vista completaPublicado el 27 de diciembre de 2017 | http://doi.org/10.5867/medwave.2017.09.7117
¿Es efectiva la profilaxis pre-exposición para prevenir infección por VIH en hombres que tienen sexo con hombres?
Is pre-exposure prophylaxis effective for preventing HIV infection in men who have sex with men?
Resumen
INTRODUCCIÓN Las altas tasas de infección por VIH siguen siendo de preocupación, especialmente para grupos de riesgo como hombres que tienen sexo con hombres. La profilaxis pre-exposición oral se ha perfilado como una alternativa de prevención. Sin embargo, aún persisten dudas en pacientes y tratantes sobre su efectividad.
MÉTODOS Para responder a esta pregunta utilizamos Epistemonikos, la mayor base de datos de revisiones sistemáticas en salud, la cual es mantenida mediante búsquedas en múltiples fuentes de información, incluyendo MEDLINE, EMBASE, Cochrane, entre otras. Extrajimos los datos desde las revisiones identificadas, reanalizamos los datos de los estudios primarios, realizamos un metanálisis y preparamos tablas de resumen de los resultados utilizando el método GRADE.
RESULTADOS Y CONCLUSIONES Identificamos seis revisiones sistemáticas que en conjunto incluyen doce estudios primarios, de los cuales seis son ensayos aleatorizados. Concluimos que el uso de profilaxis pre-exposición oral disminuye la probabilidad de infección por VIH en hombres que tienen sexo con hombres, tiene pocos o nulos efectos adversos, y se trataría de una medida con un buen balance entre beneficios, riesgos y costos.
Problema
Pese a un mayor conocimiento por parte de la comunidad sobre el VIH, las tasas de infección se mantienen en aumento. Uno de los mayores grupos de riesgo son los hombres que tienen sexo con hombres, por lo que se requieren mejores estrategias para su prevención. El uso de medicamentos antirretrovirales en individuos no infectados, o profilaxis pre-exposición (PrEP), se ha perfilado como una herramienta promisoria para la prevención en individuos de alto riesgo de contagio con VIH. La combinación más usada de PrEP ha sido emtricitabina con tenofovir disoproxil fumarato oral, y en menor medida tenofovir disoproxil fumarato solo. A pesar de la aprobación del primero como PrEP por parte de la FDA en 2012, tratantes y pacientes aún persisten con dudas sobre su efectividad y seguridad.
Metodos
Para responder esta pregunta utilizamos Epistemonikos, la mayor base de datos de revisiones sistemáticas en salud, la cual es mantenida mediante búsquedas en múltiples fuentes de información, incluyendo MEDLINE, EMBASE, Cochrane, entre otras. Extrajimos los datos desde las revisiones identificadas y reanalizamos los datos de los estudios primarios. Con esta información, generamos un resumen estructurado denominado FRISBEE (Friendly Summaries of Body of Evidence using Epistemonikos), siguiendo un formato preestablecido, que incluye mensajes clave, un resumen del conjunto de evidencia (presentado como matriz de evidencia en Epistemonikos), metanálisis del total de los estudios cuando sea posible, tablas de resumen de resultados con el método GRADE, y tabla de otras consideraciones para la toma de decisión.
|
Mensajes clave
|
Acerca del conjunto de evidencia para esta pregunta
|
Cuál es la evidencia |
Encontramos seis revisiones sistemáticas [1],[2],[3], |
|
Qué tipo de pacientes incluyeron los estudios* |
Los pacientes incluidos en los ensayos fueron hombres que tienen sexo con hombres, no infectados por VIH, mayores de 18 años, y considerados de alto riesgo de infección por VIH (por historia de alto número de parejas sexuales, relaciones sexuales sin uso de preservativo, relaciones sexuales con personas con enfermedades de transmisión sexual, o relaciones sexuales a cambio de dinero). |
|
Qué tipo de intervenciones incluyeron los estudios* |
Todos los ensayos evaluaron el uso de PrEP oral en conjunto con prevención estándar (incluyendo educación y provisión de preservativos). Cinco evaluaron el uso de emtricitabina/tenofovir [11],[14],[20],[23],[29] y uno el uso de tenofovir solo [25]. Cinco ensayos evaluaron contra placebo [11],[14],[23],[25],[29] y tres contra un grupo de no tratamiento [14],[20],[25]. Cinco usaron PrEP de forma diaria [11],[14],[20],[25],[29] y uno de forma intermitente, antes y después de tener relaciones sexuales [23]. Un ensayo usó como cointervención una terapia conductual [14] y un ensayo fue pragmático y abierto [20]. Uno incluyó un pequeño grupo de mujeres [29], pero dado que la gran mayoría de los participantes eran hombres que tenían sexo con hombres se decidió incluir dentro del análisis. |
|
Qué tipo de desenlaces midieron |
Los principales desenlaces analizados fueron la tasa de infección por VIH y efectos adversos. Desenlaces analizados de forma cualitativa incluyeron cambios en conductas de riesgo, principalmente uso de preservativo objetivado por entrevistas [14],[25] o indirectamente por incidencia de otras enfermedades de transmisión sexual [20]. El seguimiento promedio de los ensayos fue de 15 meses, con un rango entre cuatro y 33 meses. |
* La información sobre los estudios primarios es extraída desde las revisiones sistemáticas identificadas, no directamente desde los estudios, a menos que se especifique lo contrario.
Cuál es la evidencia
Resumen de los resultados
La información sobre los efectos de PrEP oral en hombres que tienen sexo con hombres está basada en seis ensayos [11],[14],[20],[23],[25],[29] que incluyeron 3974 pacientes.
No fue posible extraer suficiente cantidad de información desde las revisiones identificadas como para reconstruir el metanálisis sobre incidencia de VIH. Por lo tanto, la información presentada se basa en los resultados del metanálisis de una revisión sistemática [1] que está basada en cuatro ensayos [11],[23],[25],[29] que incluyeron 3371 pacientes.
La información sobre eventos adversos está basada en tres ensayos [11],[14],[25] cuyos datos eran reutilizables desde las revisiones sistemáticas e incluyeron a 2957 pacientes. La información sobre cambios en uso de preservativo está basada en tres ensayos [14],[20],[25] que compararon contra no tratamiento, cuyos datos fueron descritos de forma cualitativa en las revisiones sistemáticas e incluyeron 1003 pacientes.
El resumen de los resultados es el siguiente:
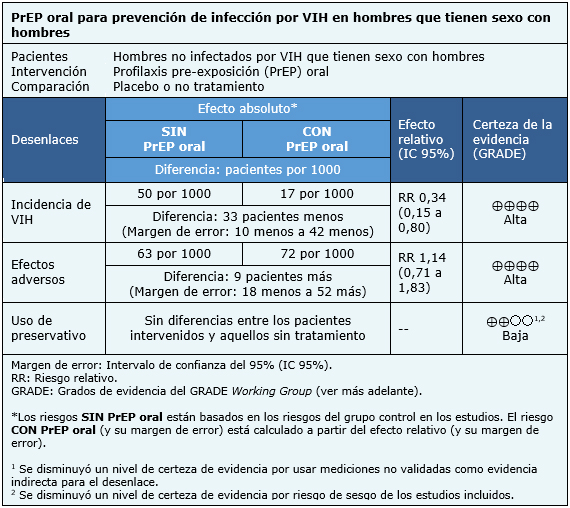
- El uso de PrEP oral disminuye la probabilidad de infección de VIH en hombres que tienen sexo con hombres. La certeza de la evidencia es alta.
- El uso de PrEP oral resulta en diferencias mínimas en efectos adversos. La certeza de la evidencia es alta.
- El uso de PrEP oral podría resultar en poca o nula diferencia en cuanto a cambios en uso de preservativo en hombres que tienen sexo con hombres. La certeza de la evidencia es baja.
| Siga el enlace para acceder a la versión interactiva de esta tabla (Interactive Summary of Findings - iSoF) |
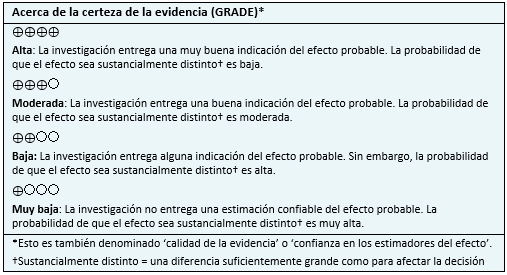
Otras consideraciones para la toma de decisión
|
A quién se aplica y a quién no se aplica esta evidencia |
|
| Sobre los desenlaces incluidos en este resumen |
|
| Balance riesgo/beneficio y certeza de la evidencia |
|
| Consideraciones de recursos |
|
| Qué piensan los pacientes y sus tratantes |
|
| Diferencias entre este resumen y otras fuentes |
|
| ¿Puede que cambie esta información en el futuro? |
|
Cómo realizamos este resumen
Mediante métodos automatizados y colaborativos recopilamos toda la evidencia relevante para la pregunta de interés y la presentamos en una matriz de evidencia.
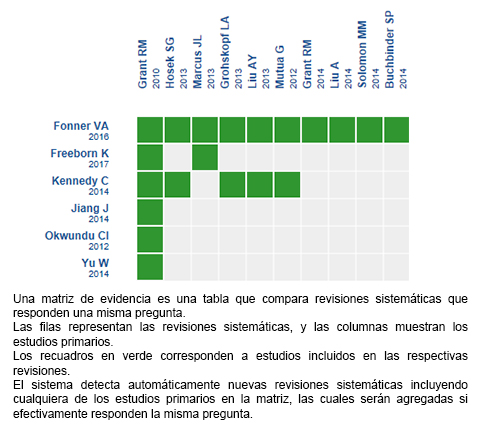
Siga el enlace para acceder a la versión interactiva: PrEP oral para prevenir infección por VIH en hombres que tienen sexo con hombres
Notas
Si con posterioridad a la publicación de este resumen se publican nuevas revisiones sistemáticas sobre este tema, en la parte superior de la matriz se mostrará un aviso de “nueva evidencia”. Si bien el proyecto contempla la actualización periódica de estos resúmenes, los usuarios están invitados a comentar en la página web de Medwave o contactar a los autores mediante correo electrónico si creen que hay evidencia que motive una actualización más precoz.
Luego de crear una cuenta en Epistemonikos, al guardar las matrices recibirá notificaciones automáticas cada vez que exista nueva evidencia que potencialmente responda a esta pregunta.
Este artículo es parte del proyecto síntesis de evidencia de Epistemonikos. Se elabora con una metodología preestablecida, siguiendo rigurosos estándares metodológicos y proceso de revisión por pares interno. Cada uno de estos artículos corresponde a un resumen, denominado FRISBEE (Friendly Summary of Body of Evidence using Epistemonikos), cuyo principal objetivo es sintetizar el conjunto de evidencia de una pregunta específica, en un formato amigable a los profesionales clínicos. Sus principales recursos se basan en la matriz de evidencia de Epistemonikos y análisis de resultados usando metodología GRADE. Mayores detalles de los métodos para elaborar este FRISBEE están descritos aquí (http://dx.doi.org/10.5867/medwave.2014.06.5997)
La Fundación Epistemonikos es una organización que busca acercar la información a quienes toman decisiones en salud, mediante el uso de tecnologías. Su principal desarrollo es la base de datos Epistemonikos (www.epistemonikos.org).
Declaración de conflictos de intereses
Los autores declaran no tener conflictos de intereses con la materia de este artículo.

