Análisis
← vista completaPublicado el 26 de octubre de 2020 | http://doi.org/10.5867/medwave.2020.09.8051
Identificación y análisis de los ensayos clínicos intervencionales planificados y registrados sobre COVID-19
Identification and analysis of ongoing registered clinical intervention trials on COVID-19
Resumen
Introducción La infección por SARS CoV-2 fue declarada pandemia el 11 de marzo de 2020. Desde entonces, se han propuesto e iniciado numerosos estudios, cuyo propósito incluye prevención, diagnóstico, manejo y tratamiento de esta nueva enfermedad.
Objetivo Identificar y categorizar los estudios intervencionales hasta mayo de 20202 relacionados con la infección por SARS-CoV-2, de acuerdo con población y ubicación geográfica (con énfasis en Latinoamérica) y comprobar si existe correlación según el propósito, fase y estado de reclutamiento de los mismos.
Metodología Se seleccionaron 1672 de los 1705 estudios disponibles en la plataforma de ensayos clínicos de la Organización Mundial de la Salud (OMS) relacionados con COVID-19. Se utilizaron las herramientas Jupyter y Python para el procesamiento y depuración de datos.
Resultados Se encontraron 1672 estudios de tipo intervencional relacionados con la infección por SARS-CoV-2. China, Estados Unidos, Irán, Francia y España son los países que participan en mayor cantidad de estudios, mientras que solamente 4,1% proviene de América Latina (mayoritariamente brasileños). Las investigaciones que se centran únicamente en adultos mayores son 28, y solamente 10 estudios se basan exclusivamente en población menor de 19 años.
Conclusión El interés mundial por esta nueva enfermedad se ve reflejado en la cantidad creciente de estudios intervencionales que se han realizado hasta la fecha. Sin embargo, los estudios analizados no abarcan proporcionalmente los grupos etarios más vulnerables, y no cuentan con una participa-ción equitativa de los países del orbe. En el caso de América Latina se agudiza esta problemática debido a las limitaciones sociales, económicas y políticas de la región. Debido a que es una enfermedad emergente, aún no existe suficiente información para poder establecer correlaciones contundentes entre las variables estudiadas. La estandarización definitiva de protocolos resulta prematura, ya que la mayoría de los estudios se encuentran en curso.
Ideas clave
- El SARS-CoV-2 es el virus responsable de la segunda pandemia del siglo XXI, convirtiéndose en un grave problema de salud pública que ha generado estragos en la población mundial.
- La investigación, si bien es una gran aliada para controlar, comprender y formular nuevas estrategias terapéuticas de las enfermedades, en este caso COVID-19; suele perpetuar patrones de exclusión de poblaciones como adultos mayores y niños. Por ello, es importante visibilizar sus diferencias y demostrar el impacto que tienen los nuevos estudios sobre ella.
- Además, existen regiones geográficas que históricamente aportan poca cantidad de estudios al total de la literatura científica mundial, como América Latina que realiza cerca del 2% de las investigaciones del orbe.
- Un posible sesgo que puede existir en este estudio es que se establece un corte arbitrario en el tiempo para incluir o no los ensayos clínicos.
- También existe sesgo en el reclutamiento de la población participante en los diferentes ensayos clínicos, donde personas que pertenecen a minorías a étnicas, que no tienen acceso a servicios de salud u otros factores, son subestimadas en estos estudios.
Introducción
COVID-19, la patología causada por el nuevo coronavirus (SARS-CoV-2), fue detectada por primera vez durante un brote en el distrito de Wuhan, China, en diciembre del año 2019. Desde su aparición, la enfermedad tuvo una rápida expansión hacia todos los continentes[1],[2], por lo que fue declarada pandemia por la Organización Mundial de la Salud (OMS) el 11 de marzo de 2020[1], con una cantidad de casos confirmados aproximados en 200 000 y superando las 8000 muertes en alrededor de 160 países para esa fecha[2].
El SARS-CoV-2 es el virus responsable de la segunda pandemia del siglo XXI, luego de la ocurrida por influenza A (H1N1) en el año 2009[1]. Actualmente, este problema de salud pública ha generado estragos en la población mundial, siendo causa de una gran cantidad de muertes, sumado a los cambios de orden económico, político y social que enfrentan los países afectados[2],[3]. Las naciones han tenido que tomar distintas medidas de confinamiento y mitigación con la intención de proteger a la población; especialmente a adultos mayores y aquellos que poseen comorbilidades como hipertensión arterial, diabetes, cardiopatía coronaria, inmunodeficiencias, entre otras. Con ello se busca prevenir no solo el desenlace fatal, sino también el colapso de los sistemas de salud[1],[4].
La investigación se ha convertido en un aliado fundamental para el adecuado control y entendimiento de la patogenia de las enfermedades, así como para la formulación de nuevas estrategias terapéuticas[3],[5],[6]. El contexto actual no ha sido la excepción, ya que se están llevando a cabo múltiples estudios relacionados con esta nueva enfermedad, los cuales abarcan temas desde el diagnóstico, manejo y tratamiento de la infección por SARS-CoV-2, hasta estrategias para prevenir su transmisión[5],[6].
Pese a la importancia de las investigaciones y en especial de estudios intervencionales durante esta pandemia, existen diferencias con respecto a los protocolos para realizarlos y el acceso a los mismos. Factores como las condiciones sociales, ambientales, políticas e inclusive el aspecto económico, juegan un papel preponderante a la hora de investigar[1].
Además, siguen perpetuándose patrones de exclusión de poblaciones como adultos mayores y niños[7], por lo que es importante visibilizar sus diferencias y demostrar el impacto que tienen los nuevos estudios sobre ellos[2]. Como indican algunos autores, las características particulares de dichos subgrupos se deben tomar en consideración a la hora de investigar ya que, debido a ellas, el impacto de la enfermedad, su prevención y tratamiento difiere con respecto a población en general[7]. Con la enfermedad causada por el SARS-CoV-2 no ha sido la excepción. Por ejemplo, en adultos mayores tiende a dar manifestaciones más graves[8], mientras que en niños los síntomas son leves; aunque se ha descrito una manifestación grave de respuesta inflamatoria[9]. Es por ello que en esta investigación se analizaron concretamente los estudios relacionados con estas poblaciones.
De igual manera, existen regiones geográficas que históricamente aportan poca cantidad de estudios al total de la literatura científica mundial, como es el caso de América Latina que realiza aproximadamente un 2% de las investigaciones del orbe[10], por lo que se estudió si esta tendencia se mantiene en esta enfermedad.
El presente artículo tiene como objetivo principal identificar y categorizar los estudios intervencionales hasta finales de mayo de 2020 relacionados con la infección por SARS-CoV-2, de acuerdo a grupo etario y ubicación geográfica (con énfasis en América Latina), además de correlacionarlos según el propósito, fase y estado de reclutamiento de los mismos.
Métodos
La presente investigación corresponde a un estudio de tipo no experimental, transversal y descriptivo. La International Clinical Trials Registry Platform (ICTRP por sus siglas en inglés), es una red que lleva registros de ensayos prospectivos, con un foro desde el cual se intercambia información y se establecen las mejores prácticas para el registro de ensayos clínicos. Se compone de registros primarios, de socios y proveedores de datos. Esta plataforma incluye los estudios inscritos en clinicaltrials.gov, Australian New Zealand Clinical Trials Registry, Brazilian Clinical Trials Registry, Chinese Clinical Trial Registry, Clinical Research Information Service, Republic of Korea, Clinical Trials Registry - India, Cuban Public Registry of Clinical Trials, EU Clinical Trials Register, German Clinical Trials Register, Iranian Registry of Clinical Trials, ISRCTN, Japan Primary Registries Network, Lebanese Clinical Trials Registry, Thai Clinical Trials Registry, The Netherlands National Trial Register, Pan African Clinical Trial Registry, Peruvian Clinical Trial Registry y Sri Lanka Clinical Trials Registry[11].
Para esta investigación se descargaron todos los estudios disponibles de forma gratuita relacionados con COVID-19 hasta el 24 de mayo de 2020, en la página https://who.int/ictrp/en/. Se corroboró, al hacer una comparación de los estudios de tipo intervencional incluidos en Cochrane y PubMed, que la base de datos de la OMS es robusta y completa. Ello, con el fin de disminuir sesgos de exclusión de investigaciones que deben formar parte del presente análisis.
La base de datos descargable consiste en un archivo csv, donde presenta cada uno de los estudios de forma tabulada. Las variables se categorizaron en treinta y tres columnas con las características de los estudios; por ejemplo: países participantes, criterios de inclusión, edad mínima y máxima de participación, resumen, entre otras. Del total de registros encontrados, se seleccionaron todos los clasificados como Interventional, interventional study o interventional clinical trial.
Se procedió a estandarizar los datos, ya que muchas columnas tenían formato numérico y nominal combinados. Por medio de la herramienta virtual libre para procesamiento de datos Jupyter, y mediante el lenguaje de programación Python, se procedió a realizar una adecuada depuración y transformación de los datos obtenidos, generando un modelo estadístico para visualización de los mismos. En el caso de las matrices de correlaciones, se utilizaron la biblioteca ScyPy y el coeficiente de correlación de Pearson. Para la definición de relación significativa, se estableció que fuera mayor a 0,5.
Para el adecuado análisis de la información se tomaron en cuenta las variables: fase del estudio (ordinal), estado de reclutamiento (nominal), propósito del estudio (nominal), países participantes (nominal), y edad mínima y máxima (ambas variables tipo discretas). Estas se escogieron debido a que su tabulación permitía una adecuada clasificación para el análisis estadístico. Se excluyeron las variables: trial ID, última actualización, título público, título científico, acrónimo, patrocinador, fecha de primer registro, fecha de segundo registro, fuente del registro, página web, condición, intervención, outcome primario, fecha esperada de resultados, fecha real de resultados, link de resultados, bridging flag, bridged type, y existencia de resultados. Se tomó esta decisión, ya que no todos los estudios contaban con dicha información, no se adecuaban al objetivo del presente estudio, y/o no eran estadísticamente valorables.
Resultados
Para la fecha del 24 de mayo del año 2020, existen 1705 estudios que cumplen con las características deseadas para la investigación. Sin embargo, en una revisión exhaustiva se pudo identificar que, según el título y las características de los estudios, cuatro de estos no se relacionan con COVID-19 y otros 29 presentan una clasificación errónea, ya que en el resumen son descritos por los autores como observacionales, por lo que se decide excluirlos del presente análisis, dejando un total de 1672 artículos.
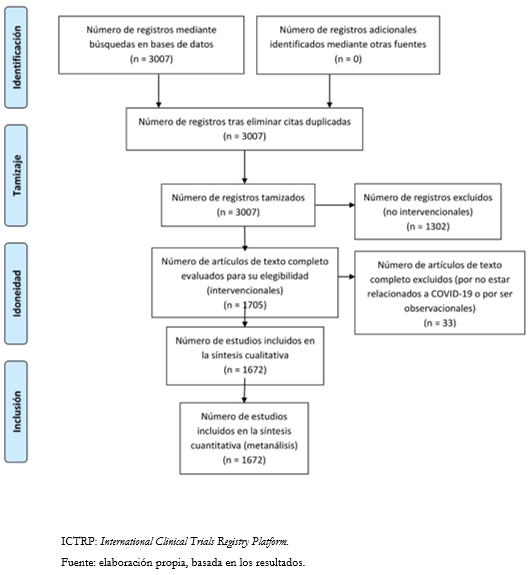 Tamaño completo
Tamaño completo Como se observa en la Tabla 1, los países que aportan más estudios son China con un total de 364, Estados Unidos con 226, Irán con 171, Francia con 128 y España con 111. El resto de los países participan en menos de 100. Los ensayos clínicos que cuentan con países de Latinoamérica son 81. Brasil participó en 30 investigaciones, México en 17, Argentina y Colombia en nueve, Cuba en cinco, Perú en cuatro, Chile en tres, y Honduras, Guyana Francesa y Ecuador en uno cada uno. Cabe destacar que en uno de ellos participaban conjuntamente Argentina y Brasil.
 Tamaño completo
Tamaño completo Los países con más estudios sin límite de edad son China, con 249 (63% del total), Irán con 84 (44%), Estados Unidos con 59 (26%), España con 42 (37%), Francia con 26 (20%) e India con 17 (31%).
Solamente cinco países de Latinoamérica se involucran en investigaciones donde los criterios de inclusión no establecen límite de edad, distribuidos de la siguiente manera: en siete estudios participa México, Colombia en cuatro, Brasil en tres, Argentina en uno y Cuba en uno. De todos estos, tres se encuentran en fase 2, cinco se encuentran en fase 5, uno en fase 1 y el resto no reporta este dato. Once de los estudios tenían como objetivo investigar sobre tratamiento o soporte, y el resto se clasificaban como otros. Cabe destacar que, de los 13 ensayos clínicos, 11 fueron realizados por los países de forma individual, mientras que dos eran multinacionales e incluían a México.
Por otra parte, 694 estudios tienen edad máxima establecida y su distribución se aprecia en la Figura 2. Dentro de estos, algunos son de población pediátrica por lo que la edad máxima va desde los 7 hasta los 130 años. La edad máxima promedio es de 74,7 años, con una mediana de 75.
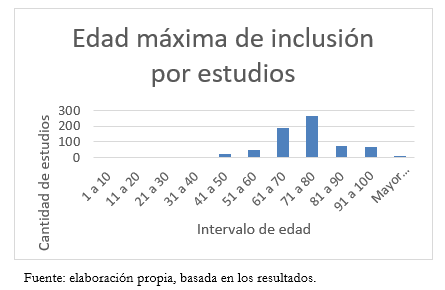 Tamaño completo
Tamaño completo No se encontró que existiera una relación significativa entre la edad límite establecida y las variables de fase del estudio, países o géneros de inclusión. En la Tabla 2 se muestra tabulada la información obtenida según fase, estado de reclutamiento y propósito. Del total de estudios analizados, 36,8% no registran información acerca de la fase en que se encuentran. Por otra parte, en la fase 2 se encuentran registrados 21,6% de los mismos, mientras que en la fase 3 16,9%. Los restantes, que comprenden 22,4%, se encuentran distribuidos entre las fases de la 0 a la 4. Al analizar otra de las variables que es el estado de reclutamiento, 799 se encuentran reclutando, mientras que 723 ya han finalizado este proceso.
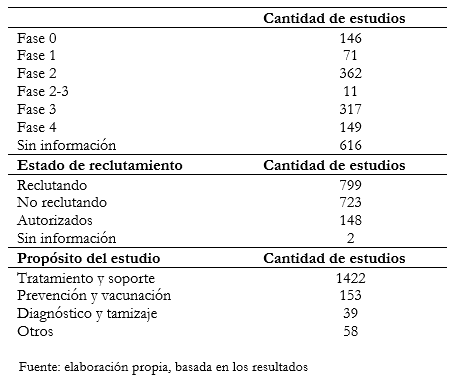 Tamaño completo
Tamaño completo En la Tabla 3 se describen únicamente los estudios que involucran a países de Latinoamérica.
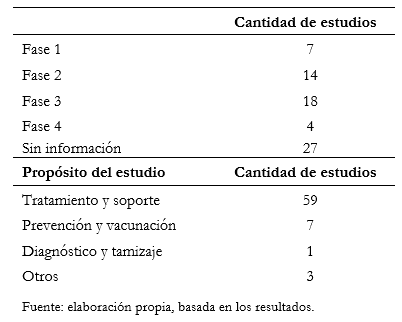 Tamaño completo
Tamaño completo Población adulta mayor
Dentro de los estudios analizados, 1581 incluyen población adulta mayor. De ellos, 896 reportan no tener límite de edad, mientras que 694 tienen una edad máxima superior a los 65 años. De los que no establecen límite de edad, 105 cuentan con la participación de este grupo etario y 9 los excluían. El resto no cuenta con dicha información.
De la totalidad de estudios, 28 se enfocan en incluir únicamente a la población mayor de 60 años, lo que equivale a 1,6% de los ensayos clínicos. De ellos uno se está realizando en América Latina, específicamente en Cuba. De esos estudios, 16 tienen como propósito la investigación en tratamiento y soporte, 11 son de prevención y uno estaba clasificado como “otro”.
Población pediátrica
Un total de 10 investigaciones incluyen exclusivamente población menor de 19 años, los cuales se llevan a cabo en Francia, China, Estados Unidos, Ghana y Canadá. Tres de ellos tienen propósito de tratamiento, dos de prevención, uno de diagnóstico y el resto no especifica. No se encontró que existiera una relación significativa entre la edad límite establecida y las variables fase, países o géneros de inclusión.
De los estudios, 136 tienen como criterio de inclusión una edad menor de 19 años, por lo que integran tanto a población adulta, como pediátrica y adolescente. Además, 36 estudios no establecen edad mínima en los criterios de inclusión. Por otra parte, 32 no cuentan con esta información.
De todos los estudios que incluyen población menor de 18 años, con respecto al propósito y fase los mismos, 81 son de tratamiento, cinco de prevención, tres de diagnóstico, cuatro “otros” y el resto no lo indica. En fase 1 hay tres, en fase 2 hay 23, en fase 3 hay 44 y en fase 4 hay cinco. En fase 0 hay 20 y el resto no lo indica.
Los países que participan en estos estudios son: Irán en 42, China en 41, India en 16, Reino Unido en 14, Estados Unidos en nueve, Egipto y Francia en siete, Italia en seis, Canadá y Japón en cinco, México y Suecia en cuatro4, Bélgica, Alemania, Corea, Países Bajos, España y Suiza en tres; mientras que Brasil, Hong Kong, Pakistán, Singapur y Taiwán en dos cada uno. Participan únicamente en un estudio Chile, Dinamarca, Guayanas Francesas, Ghana, Malasia, Nueva Zelanda, Noruega, Senegal, Sudán y Tailandia.
Discusión
Según múltiples autores, los estudios experimentales constituyen la metodología ideal en la investigación. Estos se establecen como el estándar de oro para evaluar tanto las intervenciones no farmacológicas, como la seguridad y efectividad de un medicamento en una población[12],[13]. De ahí radica la importancia de categorizar las investigaciones de este tipo y más aún cuando tratan de una patología pandémica[3].
Durante la recopilación de datos, se logró determinar que se han publicado estudios de tipo intervencional en 78 países, de los cuales 14 pertenecen al continente americano, 23 a Asia, 32 a Europa, ocho a África y uno a Oceanía. Según los resultados obtenidos, los tres países con más ensayos clínicos de este tipo son China, Estados Unidos e Irán. China posee la mayor cantidad de estudios registrados al ser el primer país en ser afectado por la enfermedad y el que se mantuvo por varios meses como el sitio con más casos a nivel mundial hasta marzo de 2020[2],[13],[14].
Para mediados de febrero en algunos países aún no se habían registrado casos de COVID-19, mientras que China ya presentaba una experiencia acumulada de casi tres meses y más de setenta mil pacientes, por lo que las investigaciones clínicas son más abundantes en esta nación[2],[13],[14]. Por otra parte, Estados Unidos es el país que históricamente ha liderado en investigación, y en 2017 era el país que más había publicado sobre brotes mundiales[15]. Además, esta nación desplazó a las demás en la cantidad de casos mundiales, ocupando hasta mayo de 2020 el primer lugar, con más de un millón y medio de casos, y con una mortalidad de 5,8%[14].
Con respecto a Irán, durante los últimos 15 años ha tenido un aumento importante en el número de artículos publicados, principalmente de literatura médica. Tiene una de las tasas de crecimiento más rápidas de las producciones científicas del mundo, con un considerable aumento de las publicaciones sobre la salud[16].
Al ser COVID-19 una patología nueva y de origen reciente, la rapidez con la que se ha avanzado en las diferentes fases clínicas de los estudios no ha sido la suficiente para llegar al término de la mayoría de los mismos. Hasta este momento, cinco meses después del inicio de la pandemia, solamente un 8,9% de los ensayos se encuentran en la fase 4. De manera similar sucede con el estado de reclutamiento, donde se evidencia que menos de la mitad (47,7%) se encuentran reclutando muestra poblacional.
Por otra parte, con respecto al propósito del estudio, 85% de ellos se ven relacionados con tratamiento y soporte de la enfermedad, y la misma tendencia se mantiene si se separa por regiones, como es el caso en América Latina. Esto es importante ya que no existe tratamiento aprobado y efectivo contra la infección por SARS-CoV-2, por lo que investigaciones que tengan como finalidad la recuperación de los enfermos resulta fundamental[17].
Situación en América Latina
Latinoamérica participa únicamente en 3,5% de los estudios analizados. De estos, 84,2% tiene como objetivo investigar sobre tratamiento y soporte de COVID-19; 10% se enfocan en prevención, 1,4% en diagnóstico y tamizaje, y 4,2% en otros temas. Con respecto a la fase, la mayoría (25%) se encuentran en fase 3, en las fases 1 y 2 un 15%, y en la fase 4 un 5,7%.
Basados en los datos obtenidos, los países latinoamericanos que más aportan estudios relacionados a COVID-19 corresponden a Brasil con 42,2%, mientras que México y Argentina contribuyen con 23,9% y 12,8% respectivamente. Esto coincide con lo que ha sucedido previamente en la región, ya que se ha caracterizado porque los países que realizan más estudios son aquellos cuya población es mayor, como lo es el caso de Argentina, Brasil y México, seguido de Colombia, Perú y Chile[16].
Para finales de marzo del presente año, en América Latina se empezó a incrementar de manera importante la cantidad de casos relacionados con la enfermedad COVID-19[14]. Es por ello que la investigación en la zona debería ser imperante. Sin embargo, históricamente esta región genera una menor cantidad de investigaciones en comparación con otras[15], patrón que se reprodujo al realizar este análisis. En consecuencia, los artículos procedentes de países latinoamericanos están poco representados en las principales revistas especializadas, debido a distintas barreras, por ejemplo, poco interés institucional, pocos incentivos, poca inversión en investigación, asuntos políticos, entre otras[18].
La problemática de América Latina se centra en el escenario que presentan muchos de los países que la componen, en donde las condiciones sanitarias, así como los problemas económicos, sociales y el accionar político (por ejemplo, la posición de subestimar el virus en Brasil y Nicaragua), constituyen factores que podrían ser desfavorables ante la expansión de la enfermedad[1].
Estas características regionales, son las que predisponen a un preludio de nuevos desafíos frente a la expansión del virus en esta región. De aquí la importancia de generar investigaciones que puedan beneficiar a la población más afectada[1],[5].
Latinoamérica tiene la ventaja de que podría aprender de las experiencias externas para evitar llegar a la situación tan drástica que se presentó en países europeos durante la actual pandemia. Aunque ya se han acatado medidas sanitarias para evitar la propagación masiva del virus y el colapso de los servicios de salud, la realización de estudios sigue siendo una medida importante que permite la generación de conocimiento necesario para optimizar las decisiones en el manejo de la pandemia[1],[2],[5].
Población adulta mayor
Se ha descrito que los ensayos clínicos de tipo intervencional que incluyen exclusivamente adultos mayores son pocos, entre 1 y 2% según algunas series, y usualmente presentan muestras pequeñas, inclusive para enfermedades que son de predominio en esta población[2].
Esto se puede explicar debido a que en los ensayos clínicos se intenta controlar tantas variables como sea posible y los adultos mayores presentan muchas comorbilidades y patologías que pueden alterar los resultados de los mismos[7]. COVID-19 no es la excepción. Pese a que la mayoría de los estudios (94%) podrían incluir adultos mayores (ya que no se establecen límites de edad o el límite es superior a 65 años), solo 1,6% de ellos analiza como subgrupo a los mayores de 60 años. Esto puede resultar en una subestimación de las consecuencias, tanto de la enfermedad como de su tratamiento en este grupo etario[19].
De los ensayos clínicos analizados, la mayoría se relacionan con prevención y tratamiento de la enfermedad. Estos son objetivos fundamentales ya que esta población presenta mayor mortalidad por COVID-19[8]. Es ampliamente conocido que la farmacocinética y farmacodinámica cambia sustancialmente, principalmente después de los 75 años[2]. Es por ello que el riesgo/beneficio de nuevos tratamientos en el manejo de COVID-19 debe ser estudiado ampliamente. Además, se deben establecer más investigaciones que tengan como objetivo principal determinar eficacia y seguridad en este subgrupo.
Con respecto a la distribución geográfica de los ensayos clínicos que incluyen adultos mayores, la mayoría de ellos se llevan a cabo en China, primer país afectado por COVID-19. También se realizan en regiones donde hasta mayo de 2020 había gran cantidad de casos, por ejemplo, Estados Unidos y España[14]. Faltan más estudios con este subgrupo en todas las regiones del mundo. Particularmente, se encontraron pocos con esta población en América Latina. Por ejemplo, hasta mediado de mayo, el país con más casos confirmados en la región era Brasil[14]. Sin embargo, pese a contar con normas éticas y regulatorias bien establecidas en el ámbito de la investigación, presenta pocos ensayos clínicos que involucran adultos mayores. Algunos autores han propuesto como limitante la demora entre el tiempo de aprobación y el inicio de los ensayos clínicos en este país[8].
En general, la población adulta mayor tiene un riesgo similar de infección por el virus SARS-CoV-2. Sin embargo, gran parte de ellos presentan comorbilidades como diabetes, hipertensión arterial, enfermedad cardiovascular o cerebro vascular. Estas, sumadas a la edad, sí les confieren un riesgo de letalidad mayor. Según algunas hipótesis, esto se da por el debilitamiento del sistema inmunológico asociado a otras enfermedades[8].
También, es la población con más alta mortalidad asociada a la enfermedad COVID-19, debido a que presentan mayor susceptibilidad de enfermar gravemente, pero una menor posibilidad de ser admitidos en una unidad de cuidados intensivos[18]. Se describe que además de complicarse con neumonía debido a COVID-19, es el subgrupo más propenso a realizar disfunción y fallo multisistémico, por lo que al abordar a un paciente adulto mayor se deben prevenir estas otras complicaciones[8].
Población pediátrica
De la totalidad de estudios analizados, solamente un 0,5% de los mismos se enfocan exclusivamente en población menor a 19 años. De estos, 30% tiene como objetivo el tratamiento de COVID-19, 20% la prevención, 10% el diagnóstico y 40% no brinda información sobre el propósito de la investigación.
Como ya es sabido, la epidemiología de COVID-19 difiere de manera significativa entre diferentes grupos etarios. Se ha demostrado que la incidencia en niños es significativamente menor que en la población general, y que esta puede variar entre 1 y 2%[20],[21], lo cual puede ser explicado debido a una menor exposición de la población pediátrica al entorno[22].
Esta baja incidencia de la enfermedad en la población pediátrica puede ser la causa de porqué muy pocos estudios se centran en este subgrupo. Sin embargo, aunque la cantidad de casos en niños es menor, estos se consideran una fuente importante de contagio[9],[15], ya que la mayoría se presentan como casos leves y asintomáticos[9]. Por lo tanto, aumentar la investigación, y más aún, incluir a dicha población en estudios de diagnóstico, es de gran importancia con el objeto de obtener datos reales de un grupo que probablemente es subdiagnosticado[9].
Además, a pesar de que la presentación clínica más frecuente es leve o asintomática, ha surgido una asociación entre la enfermedad de Kawasaki y COVID-19 en niños; específicamente en quienes tienen una respuesta inflamatoria robusta, que cursa generalmente de manera grave o fatal[23],[24]. Ante este emergente síndrome, es importante que se puedan proponer y realizar más investigaciones donde se aborde el posible tratamiento de estos pacientes y su evolución.
Del total de estudios que incluye población menor a 18 años (136), 30% no indican en qué fase del mismo se encuentran; 32,3% se encuentran en fase 3; 16,9% en la fase 2 y el 20,5% restante se encuentra distribuido entre las fases 0, 1 y 4.
Por otra parte, todas las investigaciones que se centran en el grupo etario analizado se realizan en países con alto índice de desarrollo humano[1], a excepción de una que se realiza en Ghana. Cabe destacar que ninguna de ellas incluye naciones de Latinoamérica[1]. A pesar de que la tasa de fecundidad latinoamericana ha disminuido en los últimos años, se ha visto que es significativamente mayor en los países más pobres de la región (hasta cinco hijos por mujer)[25], por lo que se puede extrapolar y considerar una densidad de población pediátrica mayor en estos países. De ahí la importancia que implica la generación de estudios en la población pediátrica en la región.
A pesar de que no ha sido documentada la trasmisión vertical como forma de contagio, se han documentado neonatos, hijos de madres infectadas, positivos por COVID-19, incluso a las 36 horas edad[26]. En el caso de los estudios analizados que incluían población pediátrica, 26,4% no establece un límite inferior de edad. Sin embargo, ninguno de ellos se ha dedicado exclusivamente a prevención, diagnóstico, manejo o tratamiento de población neonatal relacionado con COVID-19. Esto puede servir como precedente para que, en el futuro, se realicen estudios en Latinoamérica, y también exclusivamente enfocados en el contagio de la infección de SARS-CoV-2 en neonatos.
Limitaciones del estudio y consideraciones a futuro
Entre los posibles sesgos que pueden existir es el hecho de que este estudio establece un corte arbitrario en el tiempo para incluir o no los ensayos clínicos. Como bien es sabido por la comunidad científica, la cantidad de información y nuevos ensayos clínicos surgen cada minuto por lo que se podría excluir involuntariamente estudios fundamentales en el transcurso de esta pandemia. Es imprescindible que este tipo de estudios sean reproducidos en el futuro, con el fin de orientar a la comunidad científica acerca de déficits de información en la investigación necesarias para el adecuado abordaje de la pandemia.
A pesar de que se utilizó una base de datos robusta, y se corroboró que tuviera los estudios registrados tanto en PubMed como en Cochrane, no se puede garantizar que la de la OMS sea completamente exhaustiva.
Durante la recolección de datos se seleccionaron las variables que se encontraban acorde a los objetivos del estudio, mientras que se eliminaron aquellas que no tenían relación con este o no podían ser estadísticamente valorables. Por estas razones, podrían generarse a futuro nuevos estudios donde pueda incluirse información que no fue tomada en cuenta para la realización del presente escrito.
Es importante destacar que la investigación y permisibilidad de realización de ensayos clínicos no es la norma en todas las regiones o países, por lo que se pueden generar errores a la hora de estandarizar protocolos de manejo y atención basados en estudios que se realizan en zonas geográficas y con características sociodemográficas distintas. Además, existe un sesgo en el reclutamiento de la población participante en los diferentes ensayos clínicos, donde personas que pertenecen a minorías étnicas, así como las que no tienen acceso a servicios de salud, o son afectadas por otros factores de orden económicos, sociales, geográficos y culturales, son subestimados en estos estudios[27].
Pocos estudios incluyen poblaciones especiales, por ejemplo, niños, adultos mayores, pacientes gestantes, en lactancia o neonatos, por lo que es importante que se incluyan ensayos clínicos con apertura a estos subgrupos o que sean específicos para los mismos, debido a que sus características difieren de la población general. Además, con respecto a los adultos mayores, aún no se cuenta con información necesaria para saber cuál será la edad máxima real, y se tendrán que realizar nuevos estudios para determinar si el porcentaje de participación será estadísticamente significativo.
Conclusiones
Gracias a esta recopilación y categorización de los estudios de tipo intervencional relacionados con COVID-19, se pudo documentar que pocos meses después del inicio de la pandemia existe un creciente interés mundial por la investigación de esta infección, aparentemente motivado por el rápido ritmo de crecimiento de la enfermedad, la alta tasa de contagio y expansión global de la misma.
Los países que tradicionalmente han presentado mayor trayectoria en investigación en el ámbito mundial son también los que han liderado en cantidad de ensayos clínicos de tipo intervencional relacionados con COVID-19 hasta mayo de 2020. Además, las naciones que han recibido un impacto fuerte por la infección de SARS-CoV-2 se caracterizan porque han aportado mayor cantidad de estudios sobre el tema de este análisis.
En el caso específico de América Latina, los países que realizan mayor investigación en el tema presentan una mayor población. Sin embargo, las investigaciones en la región son escasas y dispersas, con una baja participación porcentual en el total mundial. Además, se evidencia una relación entre las limitaciones sociales, políticas y económicas de Latinoamérica y la ejecución de investigaciones durante esta pandemia.
Con respecto a poblaciones especiales como niños y adultos mayores, la cantidad de ensayos clínicos no se distribuye de manera representativa. Son pocos los estudios exclusivos en estos grupos etarios, con el agravante de que, debido a sus características fisiológicas particulares, los resultados que se obtienen en población general no se pueden extrapolar.
Debido a que es una enfermedad emergente, muchos de los estudios se encuentran aún en curso, por lo que establecer conclusiones y protocolos definitivos según medicina basada en la evidencia y acorde con los resultados de las investigaciones resulta prematura. Se deben realizar más análisis de corte epidemiológico y estadístico para definir su impacto real.

