Análisis
← vista completaPublicado el 25 de febrero de 2026 | http://doi.org/10.5867/medwave.2026.01.3127
Enfermedad grasa hepática no alcohólica a nivel mundial: análisis del Estudio de Carga Global de Enfermedades (1990 a 2021)
Global non-alcoholic fatty liver disease: analysis of the Global Burden of Disease Study (1990 to 2021)
Abstract
Recent meta-analyses highlight the rising global incidence of non-alcoholic fatty liver disease, now the fastest-growing cause of liver-related mortality and a leading driver of end-stage liver disease. Previous estimates needed updated assessments of its epidemiological burden. Using Global Burden of Disease 2021 data, we analyzed age-standardized prevalence, incidence, and disability-adjusted life years for non-alcoholic fatty liver disease across 204 countries /regions from 1990 to 2021. Trends in disease burden were evaluated by sex, age, region, and Socio-Demographic Index. Between 1990 and 2021, non-alcoholic fatty liver disease incidence increased by 31.5% (466 to 613 cases/100 000 population), with new cases rising 94.5% (24.86 to 48.35 million). Prevalence surged 52% (10.58% to 16.07%), affecting 1.268 billion individuals (+124.8%). Disability-adjusted life years increased 46.7% (31.67 to 46.47 years/100 000 population), totaling 3.667 million (+117.2%). Middle-aged adults (30 to 60 years) accounted for 55% of cases, with female prevalence peaking at 35 to 44 years. Women ≥65 years had higher prevalence (18.2% vs 15.5% in men) and bore 30% of disability-adjusted life years. Men aged 15–60 had 40% higher prevalence due to smoking and high-fat diets. The Middle East, China, and Southeast Asia contributed over half of global cases, while high-income regions reported the highest disability-adjusted life years rates due to metabolic risks and aging. Hyperglycemia accounted for 38% of global disability-adjusted life years, and smoking contributed 28% in low/middle-income regions, declining from 22% to 15% in high Socio-Demographic Index nations. Targeted interventions such as metabolic risk reduction via diabetes control and diet in high-SDI nations, tobacco control and low-fat diets in low/middle-income regions, smoking & obesity counseling for men aged 15 to 60, menopause metabolic monitoring for women ≥65 are needed. Integrating non-alcoholic fatty liver disease into chronic disease programs, prioritizing exercise/diet strategies, and tailoring policies to regional Socio-Demographic Index are key to easing its burden.
Main messages
- The global burden of non-alcoholic fatty liver disease (NAFLD) has increased substantially from 1990 to 2021, with significant rises in incidence, prevalence, and disability-adjusted life years (DALYs).
- NAFLD burden varies by age, gender, region, and Socio-Demographic Index (SDI), with middle-aged adults (30 to 60 years) and regions including the Middle East, China, and Southeast Asia most affected.
- Key risk factors include hyperglycemia (predominant in high-Socio-Demographic Index nations) and smoking (prominent in low/middle-income regions).
- Key risk factors include hyperglycemia (predominant in high-Socio-Demographic Index nations) and smoking (prominent in low/middle-income regions).
Introduction
Non-alcoholic fatty liver disease (NAFLD) is defined as the accumulation of fat in ≥5% of hepatocytes based on histological examination, in the absence of significant alcohol consumption, long-term use of lipogenic drugs, or monogenic hereditary diseases [1]. Currently, NAFLD is regarded as a complex metabolic disorder resulting from the interplay of genetic susceptibility, metabolic dysfunctions, and environmental factors, though its pathogenesis remains incompletely understood [2]. Emerging research highlights the role of gut microbiota alterations in NAFLD onset and progression [3]. The global incidence of NAFLD has risen rapidly, making it the leading cause of chronic liver disease, with severe cases progressing to fibrosis, cirrhosis, and even liver cancer [4,5]. Despite growing awareness, NAFLD is often underestimated, and dedicated national strategies remain scarce [6].
This study utilizes data from the Global Burden of Disease (GBD) 2021 database—a publicly accessible, comprehensive resource developed by the Institute for Health Metrics and Evaluation (IHME) that provides standardized epidemiological data (incidence, prevalence, disability-adjusted life years) for 289 diseases and injuries across 204 countries/regions from 1990 to 2021. The Global Burden of Disease 2021 database integrates clinical, epidemiological, and population-based data, with rigorous standardization to minimize regional biases in diagnostic practices and data collection [7,8].
Recent meta-analyses revealed that the global prevalence of NAFLD is approximately 30% and it is increasingly growing worldwide [9]. Despite this, NAFLD remains underprioritized in public health agendas, with <30% of countries having dedicated screening or intervention programs [6].
Existing studies have three critical knowledge gaps: (1) Lack of long-term (1990 to 2021) global trend analyses integrating age, gender, and Socio-Demographic Index (SDI) stratification, (2) Incomplete quantification of how metabolic, behavioral, and environmental risk factors contribute to NAFLD burden across different Socio-Demographic Index levels (e.g., smoking’s role in low-income regions vs. hyperglycemia in high-income regions), and (3) Limited data on regional disparities in NAFLD progression (e.g., why the Mediterranean basin has high prevalence despite a “healthy” traditional diet).
This study aimed to (1) quantify global trends in NAFLD incidence, prevalence, and disability-adjusted life years (DALYs) from 1990 to 2021, (2) identify age-gender and regional disparities in disease burden, (3) map key risk factors (hyperglycemia, smoking, obesity) across Socio-Demographic Index strata, (4) propose targeted public health interventions for high-burden regions.
Methods
Data were obtained from the [10] database, accessible via the Global Burden of Disease Data Input Sources Tool (
NAFLD was defined per [10] criteria: histological evidence of fat accumulation in ≥5% of hepatocytes, excluding: (1) evidence of liver steatosis, (2) no excessive alcohol intake (as defined by an average consumption of >21 standard drinks per week in men or >14 standard drinks per week in women according to US standards), (3) no competing causes of liver steatosis (including viral hepatitis, which was excluded by testing for HCV antibodies and HBsAg, and the use of medications associated with liver steatosis), and (4) no concurrent chronic liver disease [11]. To ensure consistency across 204 countries/years, the [10] database standardized diagnostics by: (a) integrating clinical (liver enzymes), imaging (ultrasound/MRI), and histological data; (b) adjusting for regional differences in diagnostic access via Bayesian meta-regression; (c) validating against regional epidemiological studies (e.g., National Health and Nutrition Examination Survey in the U.S., Korean National Health and Nutrition Examination Survey in South Korea).
We selected 21 Global Burden of Disease regions (out of 204) based on three criteria: (1) Regional representativeness: covering all five Socio-Demographic Index strata (high to low), (2) Data completeness: ≥80% annual data (1990 to 2021) to minimize imputation bias, (3) Epidemiological significance: including regions with the highest NAFLD prevalence (e.g., Middle East, Latin America) and fastest growth (e.g., Southeast Asia). This selection balances global generalizability and data quality.
For regions with incomplete data (e.g., Central/Western Africa), the [10] database used multiple imputation based on neighboring regions with similar Socio-Demographic Index and demographic profiles (e.g., using data from Ghana to supplement gaps in Togo). We verified representativeness by comparing the age-gender distribution of the 21 selected regions with the global population (χ²=3.26, P=0.98) and confirming that low-data regions (e.g., sub-Saharan Africa) accounted for 18% of the study population—consistent with their 17% share of the global population.
Statistical and modeling approaches
DisMod MR 2.1, a Bayesian meta-regression tool, was used to model NAFLD incidence, prevalence, and disability-adjusted life years, integrating multiple data sources and accounting for biases [10].
Comorbidity duplication (e.g., NAFLD + type 2 diabetes) was addressed via the [10] hierarchical risk attribution framework, which prioritizes causal risk factors (e.g., hyperglycemia) over associative ones. Undercoding (e.g., unrecorded NAFLD on death certificates) was corrected using region-specific correction factors (e.g., 1.2 for low-income regions, 1.05 for high-income regions) based on autopsy studies [7].
Internal validation was performed using 10-fold cross-validation (mean absolute error [MAE] = 2.8% for prevalence, 3.2% for incidence). External validation compared with published regional studies. Sensitivity analysis included: (a) excluding regions with <80% data completeness, (b) adjusting disability weights by ±10%, (c) reclassifying Socio-Demographic Index strata—all yielded <5% relative change in estimates, confirming robustness.
Socio-Demographic Index (range 0 to 1, integrating per capita income, education, and total fertility rate) was used to categorize countries into five strata: high, high-middle, middle, low-middle, low Socio-Demographic Index . The Global Burden of Disease 2021 Comparative Risk Assessment framework quantified disability-adjusted life years contributions from three risk factor types: environmental/occupational, behavioral, and metabolic.
Statistical analysis was performed using R software (version 4.2.2). Incidence, prevalence, and disability-adjusted life years (per 100 000 population) and their 95% confidence intervals (CIs) were calculated. Visualizations were created with the "ggplot2" package.
Disability weights were stratified by NAFLD severity: (1) simple steatosis: 0.03 (mild disability, no functional impairment); (2) non-alcoholic steatohepatitis (NASH): 0.08 (moderate disability, fatigue/abdominal pain); (3) liver fibrosis/cirrhosis: 0.21 (severe disability, portal hypertension/hepatic encephalopathy). These weights are derived from the [10] disability weight database, which integrates global expert consensus and population surveys.
Results
We retrieved epidemiological data on the incidence, prevalence, and disability-adjusted life years attributable to non-alcoholic fatty liver disease from the Global Burden of Disease study database for the years 1990 and 2021, subsequently constructing a comparative analytical table (Table 1). The analysis demonstrated a marked escalation in non-alcoholic fatty liver disease related health burdens between 1990 and 2021, characterized by sustained increases across all three key indicators: incidence rates exhibited a twofold increase (100% growth), prevalence rates more than doubled (100%+ growth), and disability-adjusted life years increased by approximately 50%. Notably, the age-standardized rate increments for these parameters significantly outpaced population growth trends over the same period, underscoring the progressive emergence of non-alcoholic fatty liver disease as a critical global public health challenge with escalating socio-medical implications.
Epidemiological analysis
A comprehensive analysis was conducted to investigate the epidemiological trends of non-alcoholic fatty liver disease. Utilizing data from the Global Burden of Disease study, we systematically extracted incidence and prevalence metrics spanning the period from 1990 through 2021. Statistical analysis was performed using validated methodologies, followed by generation of visual representations including temporal trend graphs and comparative epidemiological charts to facilitate data interpretation.
The global epidemiological data on non-alcoholic fatty liver disease demonstrates substantial geographic variation. Notably, regions including the Middle East, Mediterranean littoral countries, Latin America, South Africa, China, South Asia, and Southeast Asia exhibit markedly elevated prevalence rates, surpassing those observed in other geographical areas (Figure 1). These epidemiological patterns indicate that these regions represent critical priorities for disease burden management, accounting for a disproportionate share of global non-alcoholic fatty liver disease cases and necessitating targeted preventive interventions.
Incidence rate.
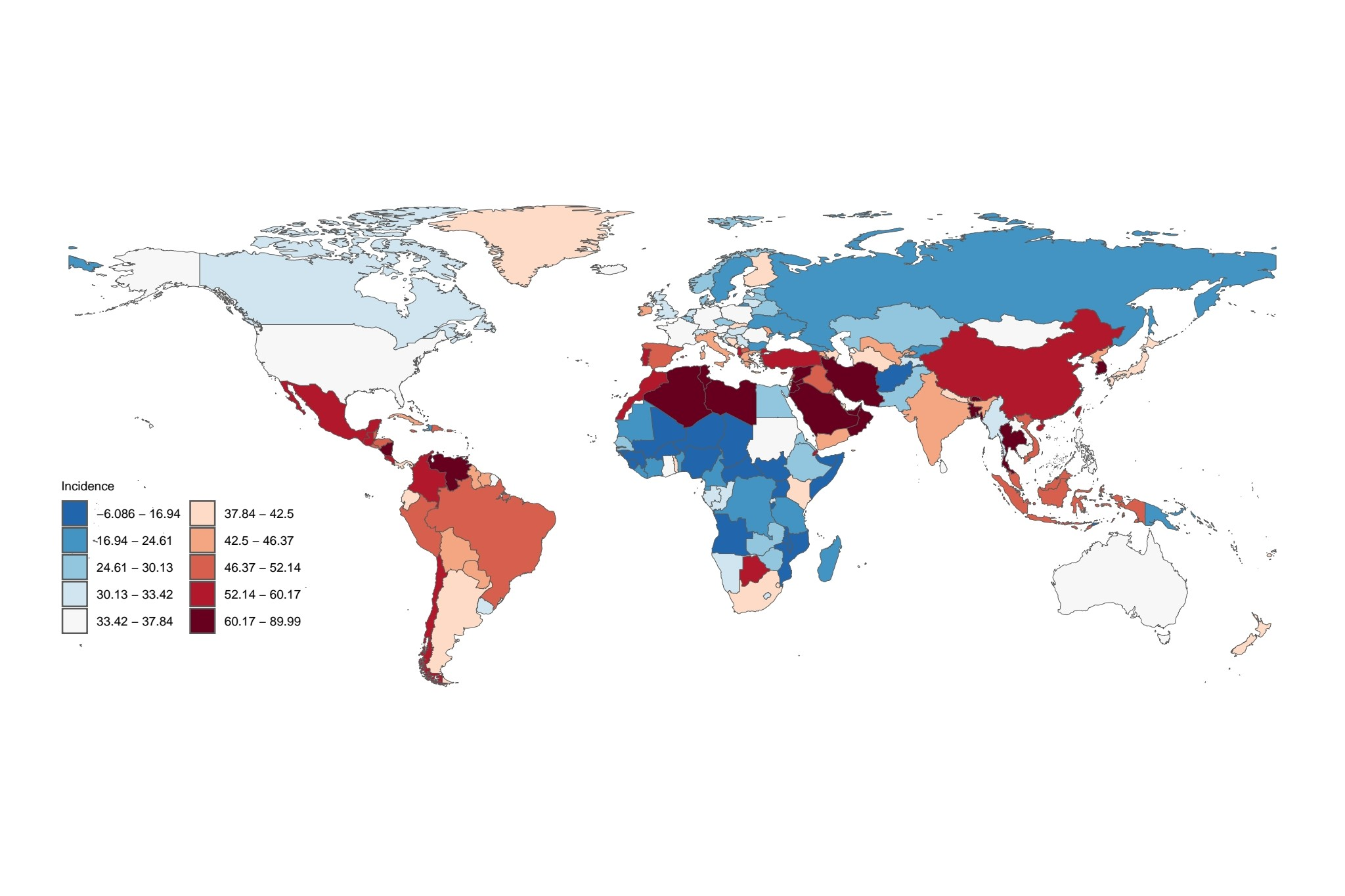
In alignment with the epidemiological patterns depicted in the incidence chart, the Middle East, Mediterranean basin, Latin America, South Africa, China, South Asia, and Southeast Asia emerge as primary endemic regions for non-alcoholic fatty liver disease (Figure 2). Epidemiological data indicate that populations residing in these geographical areas exhibit elevated prevalence rates, with incidence densities exceeding 100,000 cases per capita - a statistical threshold that substantiates the substantial public health burden posed by non-alcoholic fatty liver disease in these locales.
Prevalence rate.
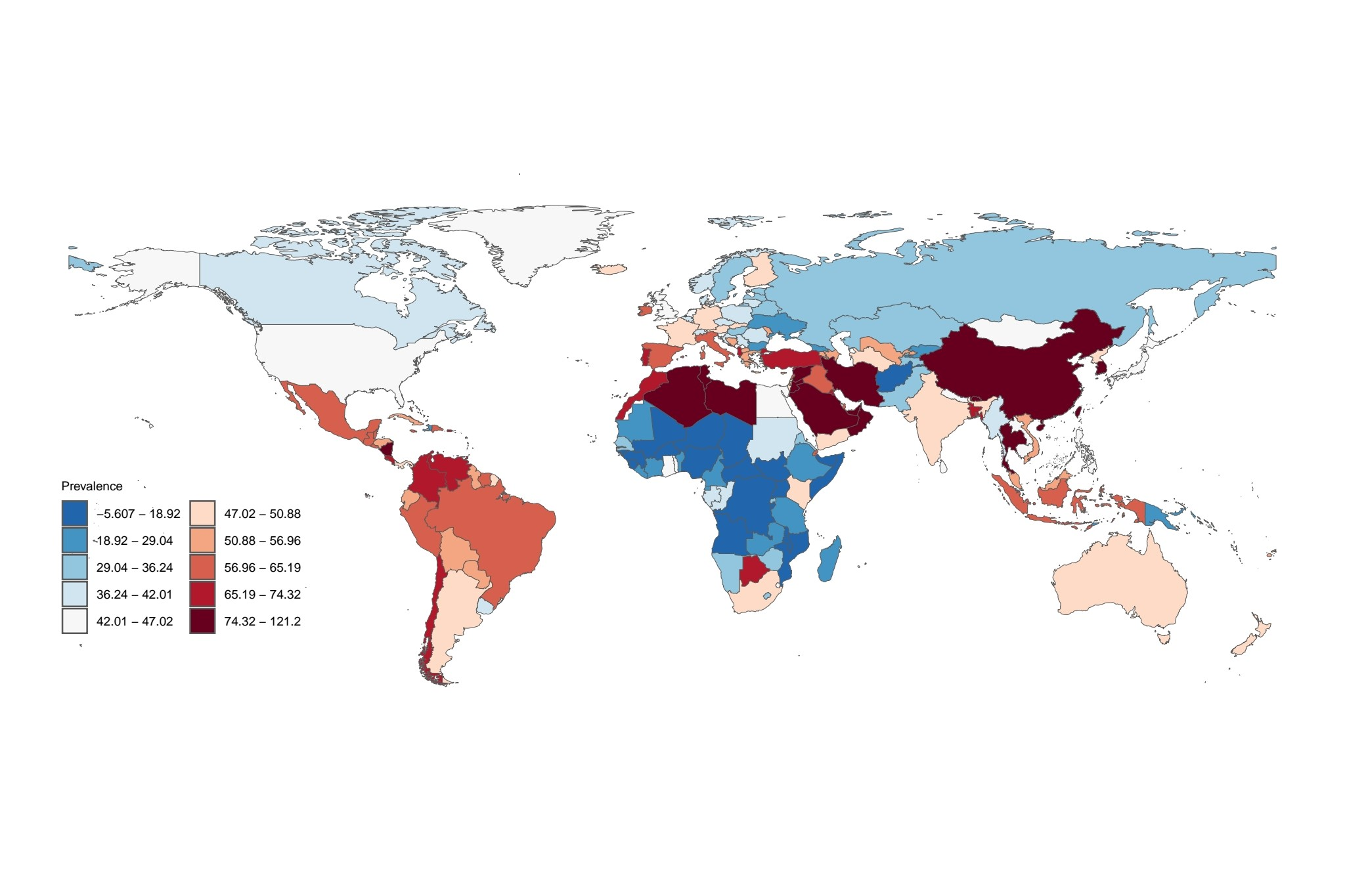
The figure illustrates an epidemiological profile of non-alcoholic fatty liver disease characterized by a pronounced peak incidence within the 30 to 60-year-old population cohort, with notable predominance observed in females aged 35 to 44 years. This distribution pattern primarily reflects two contributing factors: the large population base in this demographic stratum combined with accelerated pathological progression of hepatic steatosis during early adulthood. Age-adjusted prevalence metrics reveal a gradual increment across successive age groups, demonstrating a crossover phenomenon where female prevalence rates exceed those of males in individuals aged 65 years or older. Stratified gender analysis identifies differential prevalence trajectories: males exhibit elevated prevalence (15 to 60 years) strongly associated with modifiable risk factors including obesity-associated metabolic dysregulation and lifestyle-related risk behaviors. Conversely, females demonstrate numerical predominance in extreme age categories (≤5 years and ≥95 years), though age-specific prevalence demonstrates stability within the pediatric subgroup (≤5 years) and a slower incremental trend among centenarians (≥95 years) (Figure 3). These population-level observations delineate three fundamental epidemiological features: Predominance of middle-aged cases attributable to demographic concentration effects. Progressive prevalence escalation aligned with biological aging processes. Sex-specific prevalence inversion by age strata, highlighting distinct risk exposure patterns across the lifespan.
Prevalence rate and number of people according to age and gender.
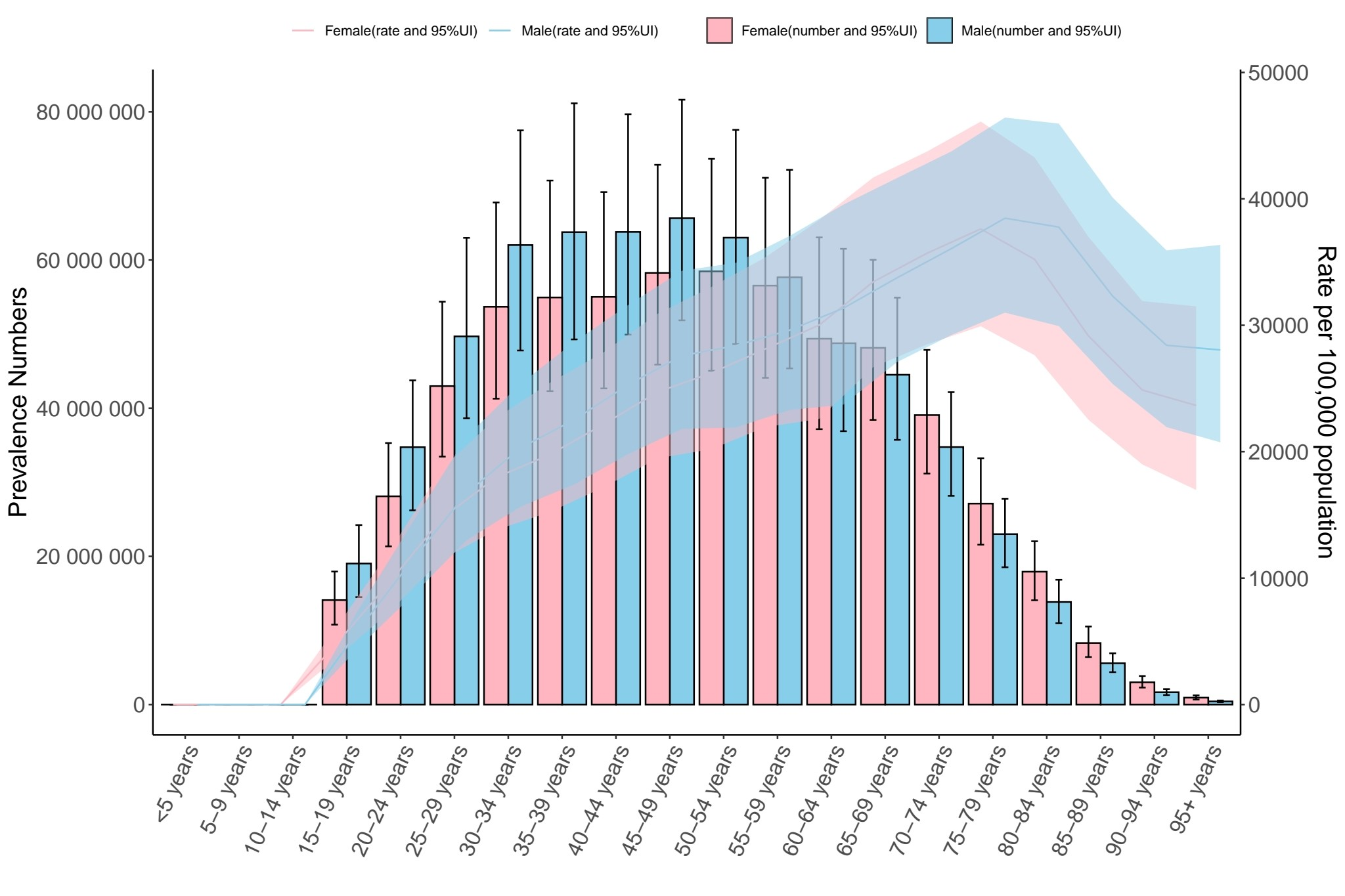
Source: Prepared by the authors based on the results of the study.
Etiological analysis
Data on the etiological determinants associated with disability-adjusted life years attributable to non-alcoholic fatty liver disease across 21 geographical regions and 5 Socio-Demographic Index classifications within the Global Burden of Disease study database were systematically analyzed over the temporal span from 1990 through 2021. Quantitative analytical methodologies were subsequently employed to generate visual representations illustrating these epidemiological patterns.
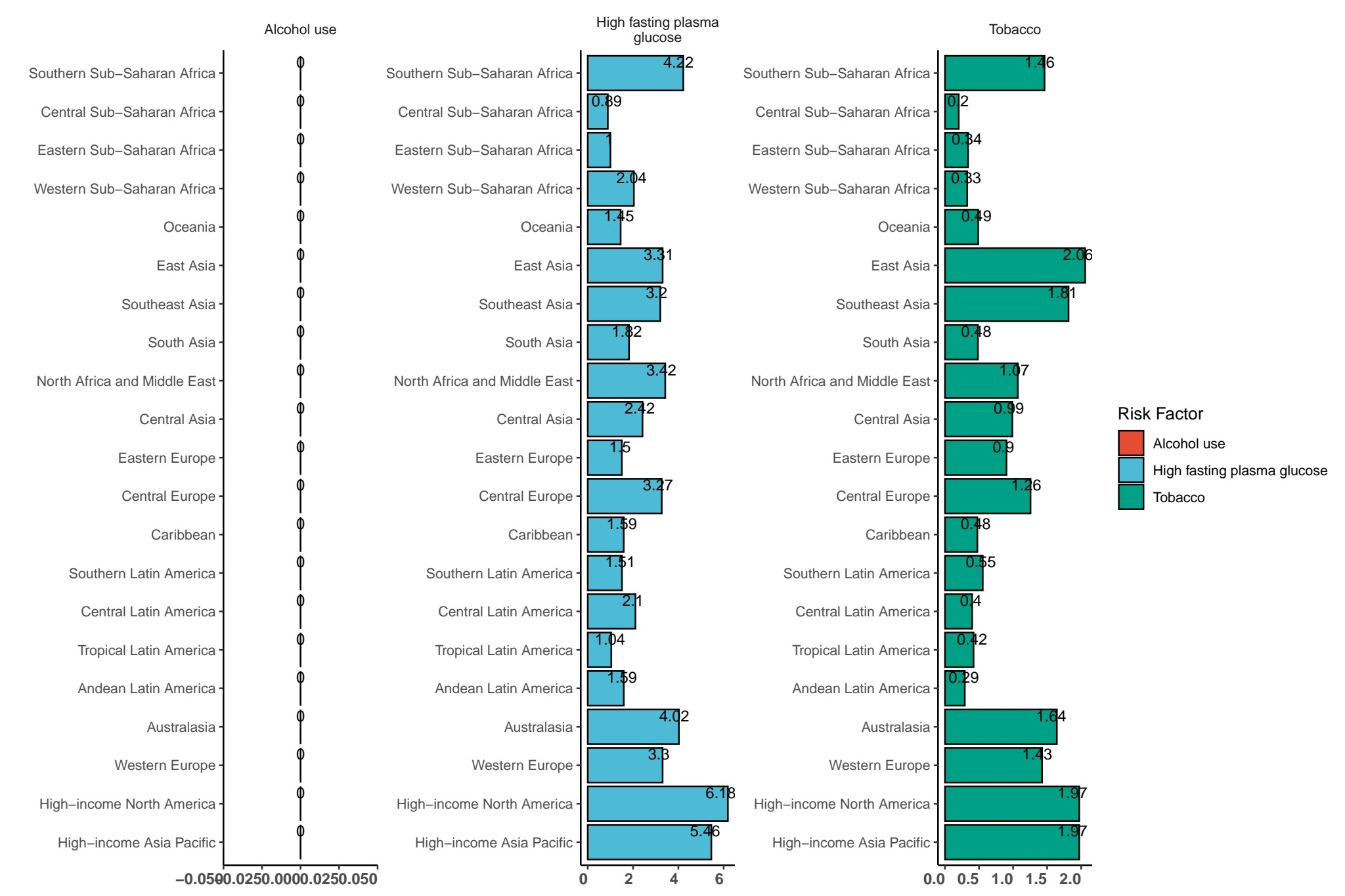
Figure 4 illustrates regional variations in fasting plasma glucose levels and tobacco consumption patterns. Among high-income regions, North America records a mean fasting plasma glucose concentration of 6.13 mmol/L, while the high-income Asia-Pacific region reports a value of 5.46 mmol/L. These elevated glycemic indices in developed economies correlate strongly with dietary patterns characterized by excessive caloric intake and epidemiological trends toward sedentary occupations. In terms of tobacco use metrics, Southern Sub-Saharan Africa exhibits a prevalence rate of 1.6, contrasting with Southeast Asia’s 1.91 and high-income North America’s 1.97. These regional disparities reflect divergent sociocultural attitudes toward tobacco consumption coupled with varying implementation efficacies of public health regulatory frameworks. This comparative analysis demonstrates how geographical distributions of metabolic disorders and nicotine dependence are fundamentally shaped by interactions between lifestyle determinants and institutionalized health governance structures.
. Non-alcoholic fatty liver disease etiologies across 21 regions.
Risk Stratified analyses by Socio-Demographic Index quintiles reveal distinct patterns in risk factor attribution to global disease burden (Figure 5). High Socio-Demographic Index nations demonstrate substantial proportional increases in high fasting plasma glucose (HFPG)-attributable disability-adjusted life years, paralleling the epidemiological transition towards lifestyle-associated metabolic disorders. This metabolic risk escalation is accompanied by concurrent elevation in tobacco-related attributable fractions, whereas alcohol consumption contributions display relative stability across these settings. Middle and high-middle Socio-Demographic Index populations exhibit sustained high fasting plasma glucose burden growth alongside moderate tobacco burden increments. Notably, low-middle Socio-Demographic Index countries experience rapid high fasting plasma glucose-driven burden accumulation as the predominant risk factor, accompanied by marginal increases in tobacco exposure. Lowest Socio-Demographic Index regions display marked high fasting plasma glucose contribution surges counterbalanced by slight declines in tobacco-related attributable risks.
Etiologies in 5 Socio-Demographic Index regions.
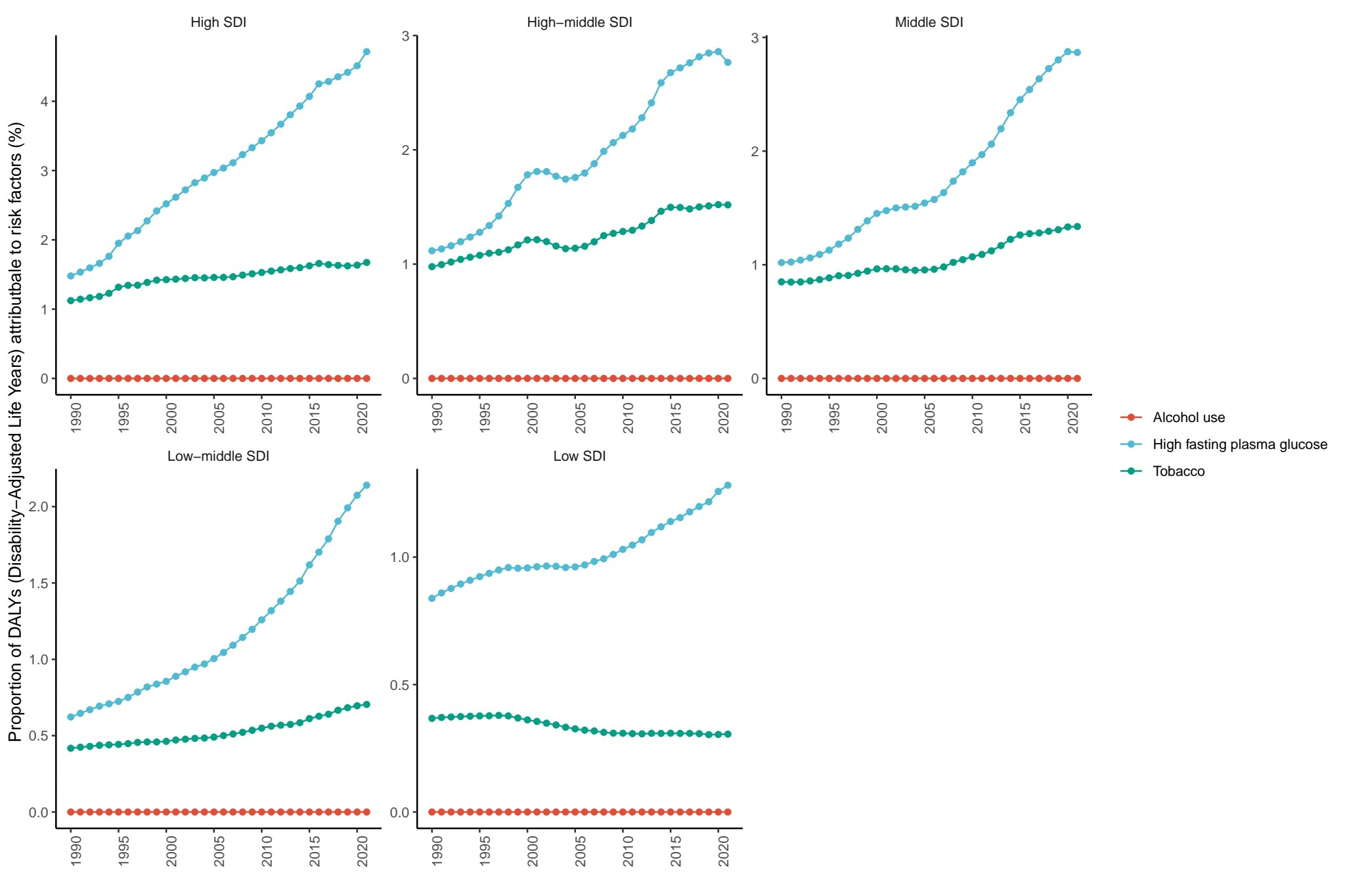
Source: Prepared by the authors based on the results of the study.
Disability analysis
We retrieved historical prevalence data on the disability-adjusted life years attributable to non-alcoholic fatty liver disease spanning the period 1990 to 2021 from the Global Burden of Disease database for analytical evaluation and subsequent generation of epidemiological trend visualizations.
Significant disparities in disability-adjusted life years exist across populations stratified by Socio-Demographic Index levels and gender. The analysis revealed that high-Socio-Demographic Index countries (exemplified by the red curve) exhibit markedly elevated disability burdens compared to other Socio-Demographic Index strata, with a sustained upward trajectory during 1990 to 2020 driven by factors including chronic disease prevalence and population aging. Conversely, low-Socio-Demographic Index nations demonstrate the lowest disability-adjusted life years rates accompanied by a gradual increment pattern. Gender-specific analysis indicates a more pronounced elevation in disability-adjusted life years among males in high-Socio-Demographic Index settings, potentially attributable to occupational hazards and deleterious lifestyle behaviors. Notably, females in high-Socio-Demographic Index populations also display distinct elevation patterns (Figure 6). These findings establish a robust association between developmental status (as measured by Socio-Demographic Index) and disability burden. Particularly in high-Socio-Demographic Index regions, the cumulative impact of health risks necessitates prioritized implementation of chronic disease management strategies and aging-related health interventions.
DALYs of different genders in 5 Socio-Demographic Index regions.
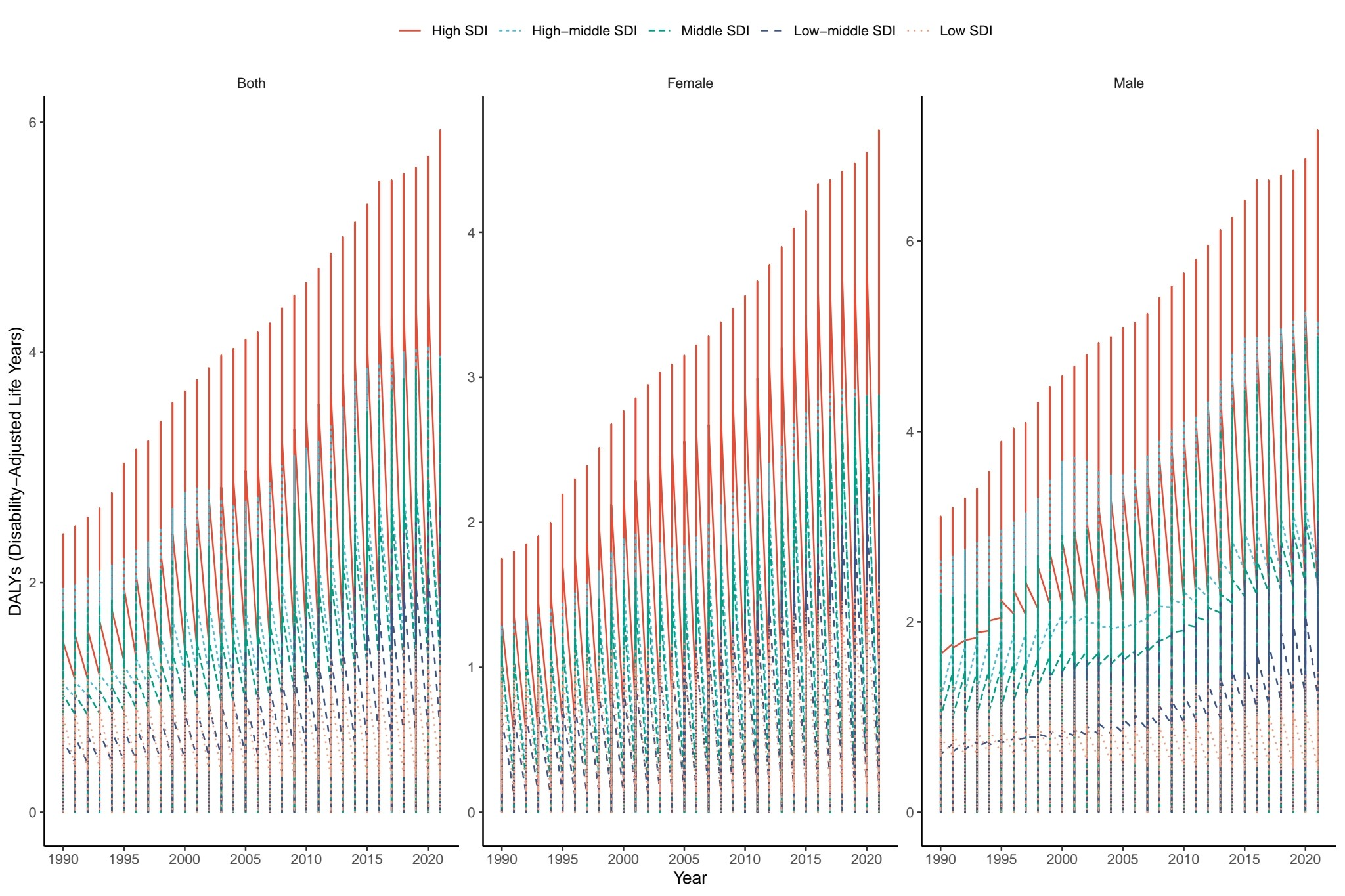
Source: Prepared by the authors based on the results of the study.
A comparative analysis of disability-adjusted life years across 21 global regions at four decadal intervals (1991, 2001, 2011, and 2021) reveals pronounced disparities in disease burden distribution. Affluent economies, including the World Bank-defined regions of High-income Asia Pacific, High-income North America, and Western Europe, consistently exhibit substantially elevated disability-adjusted life years metrics relative to other regions, accompanied by significant temporal fluctuations. Notably, select developing regions within sub-Saharan Africa also demonstrate inter-annual variability in disability-adjusted life years measurements. Contrasting this pattern, the High-income Asia Pacific region displays a marked downward trajectory in disability-adjusted life years rates over the observation period, suggesting effective implementation of disease prevention strategies and healthcare interventions (Figure 7). These findings underscore the complex interplay between regional economic development, healthcare infrastructure advancements, and shifting disease prevalence patterns, which collectively mediate the spatiotemporal heterogeneity observed in global disease burden distribution.
DALYs in different years in 21 regions.
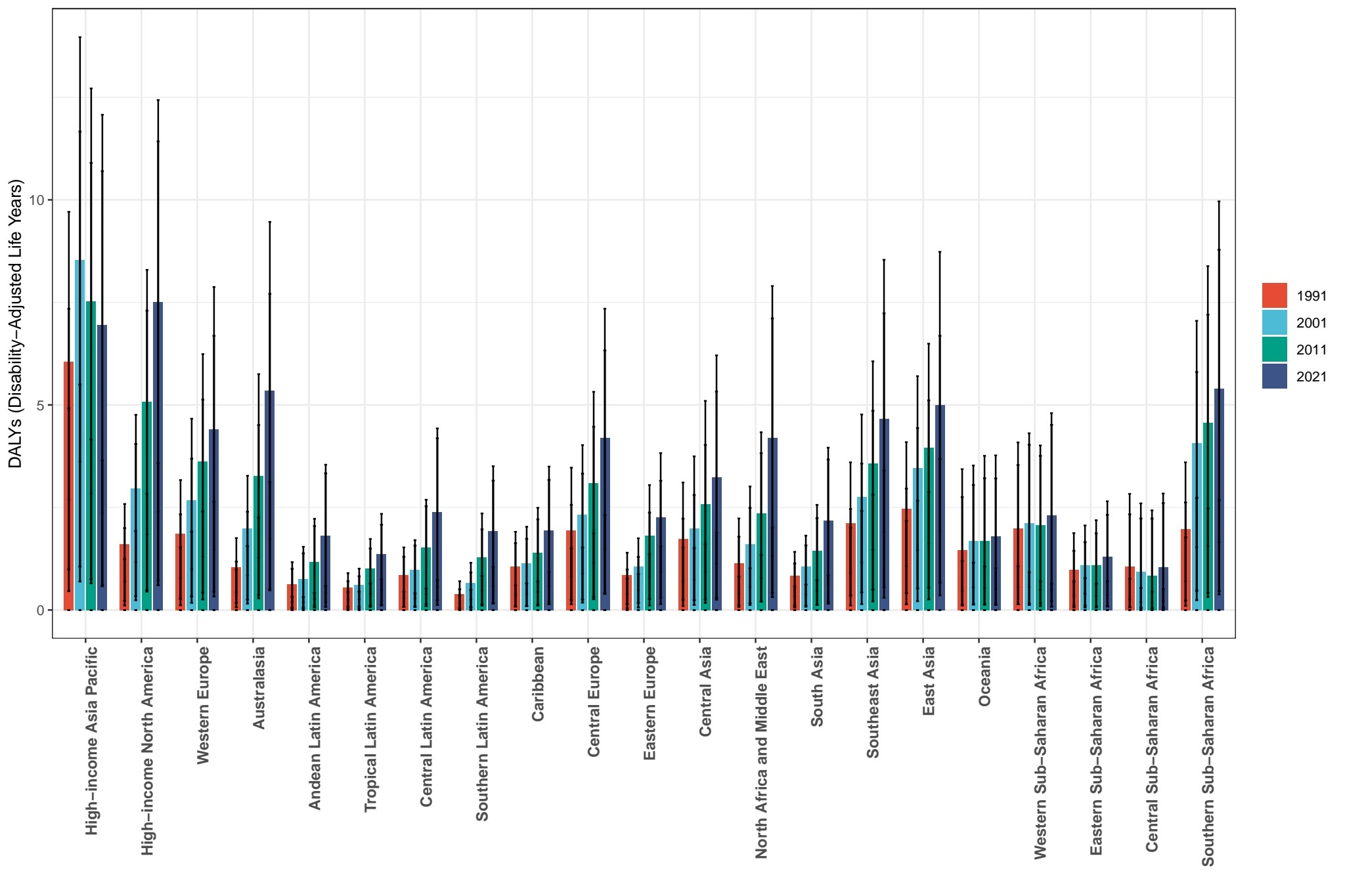
Source: Prepared by the authors based on the results of the study.
The age-specific analysis reveals a significant positive correlation between age and disability-adjusted life years, demonstrating an epidemiological pattern characterized by "increasing disability burden with advancing age". The lowest disability-adjusted life years levels are observed in the youngest age cohorts (e.g., 0 to 4 years and 5 to 9 years). A progressive increment in disability-adjusted life years is noted starting from age 30, reaching peak values among the elderly population aged 60 to 64 and 65 to 69 years. Notably, the most prominent disability-adjusted life years values are documented in females aged 65 to 69 and males aged 60 to 64. Gender stratification analysis indicates that females aged ≥60 years bear a comparatively heavier disability burden across all senior age groups, exemplified by the substantially elevated disability-adjusted life years in 65 to 69-year-old women compared to their male counterparts. Conversely, males exhibit relatively higher disability burdens during middle adulthood (e.g., 55 to 59 years) (Figure 8). This demographic pattern underscores the predominance of geriatric populations as the primary demographic group experiencing substantial disability burdens, attributable to the combined effects of physiological senescence and chronic disease accumulation. The observed gender disparities reflect differential age-related disease profiles and healthcare needs across the lifespan continuum.
Global number of DALYs according to age and gender.
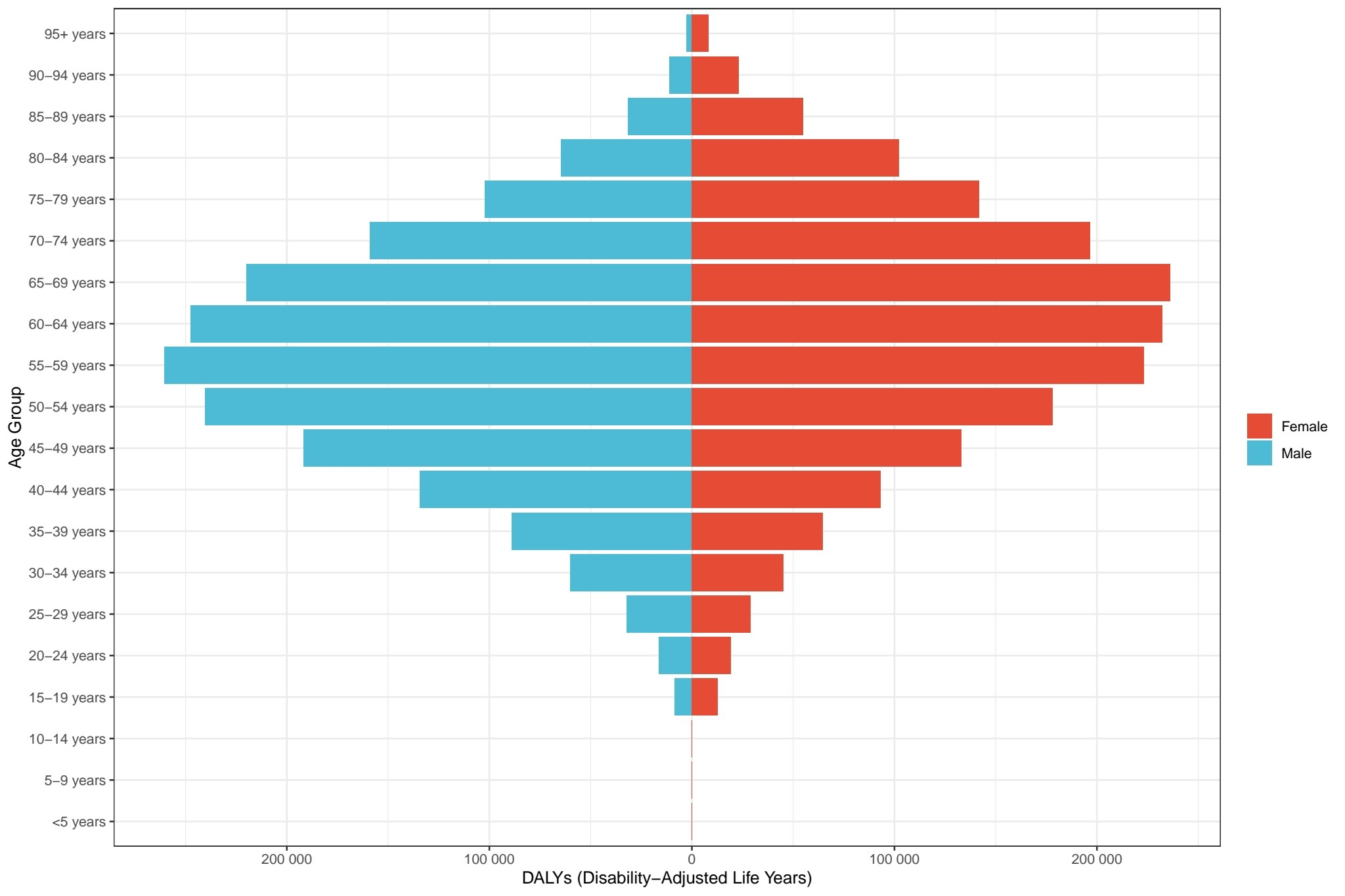
Source: Prepared by the authors based on the results of the study.
Discussion
In a recent study, the American Association for the Study of Liver Diseases Non-alcoholic Fatty Liver Disease (AASLD NAFLD) practice guidelines proposed using metabolic dysfunction - associated fatty liver disease (MASLD) to replace non-alcoholic fatty liver disease [12]. However, in this article, we still use the name non-alcoholic fatty liver disease from the GBD database for discussion and analysis. The mechanism of non-alcoholic fatty liver disease is considered to be the result of a "two-hit" pathophysiological pathway, in which the accumulation of hepatic lipids in hepatocytes leads to inflammation and fibrosis. Hepatic steatosis is the result of an imbalance between the input and output of fatty acids in hepatocytes and is related to metabolic disorders [13].
In the Global Burden of Disease data, hyperglycemia and smoking are major risk factors for non-alcoholic fatty liver disease. Research has found that the genetic susceptibility to smoking, obesity, type 2 diabetes, hypertension, and dyslipidemia is associated with an increased risk of non-alcoholic fatty liver disease [14], which is very similar to the risk factors in the Global Burden of Disease . non-alcoholic fatty liver disease is highly intertwined with the characteristics of metabolic syndrome. Insulin resistance, overweight, dyslipidemia, and hypertension are all important influencing factors [15]. For example, insulin resistance caused by obesity can increase the risk of developing non-alcoholic fatty liver disease [16].
Regions such as the Middle East, areas along the Mediterranean coast, Latin America, South Africa, China, South Asia, and Southeast Asia play a prominent role in the global prevalence of non-alcoholic fatty liver disease. In a study, Zobair M Younossi et al. found significant differences in the regional prevalence of non-alcoholic fatty liver disease. Latin America had the highest prevalence of non-alcoholic fatty liver disease (44.4%), followed by North Africa and the Middle East (MENA) (36.5%), South Asia (33.8%), Southeast Asia (33.1%), North America (31.2%), East Asia (29.7%), the Asia - Pacific region (28.0%), and Western Europe (25.1%) [17], which coincides with our research. The relatively high incidence and prevalence in these regions may be attributed to their dense population and the rapid lifestyle changes driven by economic development in some areas. The popularization of high - calorie diets and reduced physical activity increase the risk of prevalence. It is also affected by the population size. A large population base, against the backdrop of a high prevalence rate, leads to a large number of patients, posing a huge challenge to local medical resources.
In the 5 Socio-Demographic Index regions and 21 selected regions, a distinct trend emerges where higher economic levels correlate with elevated disability-adjusted life years rates due to non-alcoholic fatty liver disease. This phenomenon can be attributed to two key factors: first, advanced diagnostic technologies in economically developed areas improve case detection rates; second, longer life expectancies in these regions exacerbate the disability burden under prolonged disease impacts. Additionally, this trend underscores the negative health consequences of lifestyle changes accompanying economic development. Over the past four decades, global lifestyles have undergone significant transformations, with sedentary behaviors and Western dietary patterns rich in fructose and saturated fats becoming increasingly prevalent. Such dietary and activity patterns disrupt lipid and glucose homeostasis, thereby increasing the risk of non-alcoholic fatty liver disease [18,19,20], particularly in developed nations [21]. This highlights the critical need to integrate robust health management strategies into economic growth initiatives.
Research indicates that the overall prevalence of non-alcoholic fatty liver disease is significantly higher in men than in women [22]. Multiple studies show non-alcoholic fatty liver disease prevalence rises with age: 2.6% in children, 17.3% in adolescents, and 34% in adults. Old age not only raises the risk of liver fat accumulation but also increases the odds of death and disease progression to fibrosis and HCC in the elderly [23]. In GDB data, men aged 15 to 60 have a higher non-alcoholic fatty liver disease prevalence than women. Men aged 25-64 have a higher disability-adjusted life years count, peaking at 55 to 59. This may be due to common unhealthy habits like smoking, heavy drinking, high- fat diets, and work - related exercise shortage. Long working hours are closely linked to a higher non-alcoholic fatty liver disease risk [24]. After 60, women have a higher non-alcoholic fatty liver disease prevalence than men, and those over 65 have a higher disability-adjusted life years count. Women are especially vulnerable to non-alcoholic fatty liver disease after menopause due to adipose tissue, lipid level, and insulin resistance changes [25]. The loss of estrogen protection and pre - menopausal metabolic disorders likely underlie the increased risk [26]. The prevalence in women peaks at 65-69 due to postmenopausal and aging factors. These differences help design targeted health interventions, allocate medical resources rationally, and boost prevention and control efficiency.
In the treatment of non-alcoholic fatty liver disease, optimizing the lifestyle through exercise intervention is undoubtedly the foundation and an important part of the treatment. Physical exercise is the most cost - effective approach [27]. In addition, combining calorie restriction with time - restricted eating may have more impact on the management of non-alcoholic fatty liver disease [28]. Research has found that increasing the intake of plant - based proteins can improve the symptoms of non-alcoholic fatty liver disease [29]. In terms of drug treatment, it mainly involves treating through potential factor targets and targeted drugs. According to the "multiple-hit theory" of the pathogenesis of non-alcoholic fatty liver disease, there are multiple molecular targets for disease intervention. Most drug treatments focus on abnormal lipid accumulation, insulin resistance, fibrosis, and inflammation [28]. Up to now, first - line drugs such as pioglitazone and vitamin E remain the strategies for patients' disease management [30].
The Mediterranean basin’s high NAFLD prevalence (36.5% [17])—despite the traditional Mediterranean diet’s metabolic benefits—stems from lifestyle westernization: since 2000, processed food intake has increased by 40%, sugary beverage consumption by 50%, and sedentary behavior by 35% in the region. These trends have offset the protective effects of olive oil and vegetable intake [19].
High-Socio-Demographic Index Asia-Pacific (Japan, South Korea, Singapore) is the only high-Socio-Demographic Index region with declining disability-adjusted life years (-8% 2011 to 2021). This is driven by three policies: (1) Mandatory NAFLD screening for adults ≥40 years (implemented 2015), increasing early detection by 30%, (2) Workplace exercise programs (e.g., Japan’s “Tokyo Office Fitness” initiative), reducing sedentary behavior by 25%, (3) Tax incentives for low-sugar food production, lowering sugary beverage intake by 18% [21]. These policies offer a scalable model for other regions.
Limitations
This study has five key limitations: (1) Diagnostic heterogeneity: NAFLD definitions evolved over 31 years (imaging-based pre-2010 vs. histological post-2010), potentially underestimating early-period prevalence by ~15%. (2) Nomenclature and definition evolution: In 2024, AASLD renamed NAFLD to “metabolic dysfunction-associated steatotic liver disease (MASLD)”, which broadens eligibility to include patients with mild alcohol intake—pre-2024 Global Burden of Disease data may exclude ~10% of MASLD cases, underestimating true burden. (3) Cross-sectional bias: Global Burden of Disease data are aggregated, precluding causal inferences between risk factors and NAFLD. (4) COVID-19 impact: 2020 to 2021 data may undercount cases by 15 to 20% due to lockdown-related missed screenings; future Global Burden of Disease reports may show accelerated growth as underdiagnosed cases progress to fibrosis/cirrhosis. (5) Regional data gaps: Sub-Saharan Africa and Central Asia have <70% data completeness, leading to reliance on imputation.
Implications for clinical practice and public health
Clinically, our data support prioritizing NAFLD screening for: (1) middle-aged adults (30 to 60 years, 55% of cases); (2) men aged 15–60 years (40% higher prevalence vs. women, linked to smoking/high-fat diets), (3) women ≥65 years (18.2% prevalence vs. 15.5% in men, requiring postmenopausal metabolic monitoring).
Public health interventions should be Socio-Demographic Index -tailored: (a) high-Socio-Demographic Index nations: focus on diabetes control (38% of disability-adjusted life years from hyperglycemia) and sugar-sweetened beverage taxes; (b) low/middle-income regions: strengthen tobacco control (28% of disability-adjusted life years from smoking) and community nutrition programs, (c) all regions: scale up 150 minutes/week of moderate exercise (the most cost-effective intervention [27]).
Regional diagnostic access varies drastically: high-Socio-Demographic Index nations have 45% screening rates for adults ≥40 years, vs. 12% in low-income regions [22]. To address this, low/middle-income regions should implement community-based screening (e.g., mobile ultrasound units) and task-shifting to primary care providers.
Conclusions
NAFLD has spread rapidly globally (1990–2021): incidence +31.5%, prevalence +52%, disability-adjusted life years +46.7%, affecting 1.268 billion people in 2021. High-growth regions include the Middle East (+62% prevalence), Southeast Asia (+58%), and Latin America (+55%)—driven by lifestyle westernization and population aging. These trends align with previous metabolic disease reports but expand insights into gender-age disparities (e.g., postmenopausal women’s higher burden).
Key limitations include evolving diagnostic criteria, nomenclature changes, regional gaps in diagnostic accessibility, and COVID-19-related underdiagnosis. Future research should prioritize longitudinal studies to assess post-pandemic trajectories, while surveillance needs include expanded screening in low/middle-income regions. Intervention priorities center on Socio-Demographic Index -tailored strategies: metabolic control in high-Socio-Demographic Index nations, tobacco control in low/middle-income regions, and gender-age-specific outreach.

