Revisión clínica
← vista completaPublicado el 12 de marzo de 2026 | http://doi.org/10.5867/medwave.2026.02.3143
Aspectos generales de rehabilitación en Síndrome de Guillain-Barré: revisión narrativa
General aspects of rehabilitation in Guillain-Barre syndrome: A literature review
Abstract
Introduction Guillain-Barré syndrome is the most common cause of acquired flaccid paralysis worldwide. Although care traditionally focuses on overcoming the acute phase, many patients continue to experience residual symptoms such as weakness, neuropathic pain, autonomic dysfunction, and fatigue. These sequelae can result in significant disability and adversely affect long-term functionality and quality of life, underscoring the importance of an early, coordinated, and multidisciplinary treatment approach.
Methods A non-systematic narrative review of the literature on functional recovery in adults with Guillain-Barré syndrome was conducted. The review included narrative and systematic review articles, original research studies, clinical practice guidelines, and expert consensus statements. The search strategy prioritized rehabilitation protocols, prognostic predictors, and interventions targeting motor, sensory, autonomic, and psychosocial domains. Articles were retrieved from PubMed, Embase, and PEDro databases, with preference given to publications from 2015 to 2025, in both English and Spanish.
Results Evidence supports the implementation of a structured rehabilitation program tailored to the patient’s clinical stage, prognostic factors, and functional goals. Interventions include motor re-education, respiratory muscle training, orthotic prescription, neuropathic pain management, and symptomatic treatment of fatigue and autonomic disturbances, all while considering the individual’s personal and social context. Prognostic tools such as the Modified Erasmus Guillain-Barré Syndrome Outcome Score and the Medical Research Council sum score are useful for designing the rehabilitation plan.
Conclusion The long-term sequelae of Guillain-Barré syndrome highlight the need for a comprehensive rehabilitation approach that encompasses physical, cognitive, and psychosocial domains. Early multidisciplinary interventions and continuity of care should be the therapeutic focus. Some rehabilitation interventions still require more evidence for their implementation in practice.
Main messages
- Guillain-Barré syndrome is a potentially disabling condition due to the wide range of impairments it can cause.
- However, there is a knowledge gap, with few high-quality methodological studies on the most effective strategies for the rehabilitation of Guillain-Barré syndrome. Similarly, there are no specific clinical guidelines on this topic.
- This article brings together the main aspects to consider in the rehabilitation process, providing general guidance for a comprehensive approach to these patients.
Introduction
Guillain-Barré syndrome is a potentially fatal disease, traditionally defined as a condition of “ascending, areflexic, symmetrical, and progressive flaccid weakness” [1]. It is the leading cause of acquired neuromuscular paralysis worldwide and is preceded, in most cases, by viral or bacterial infections (e.g., Campylobacter jejuni, cytomegalovirus, or, more recently, SARS-CoV-2) [1,2,3]. The pathogenesis of Guillain-Barré syndrome is thought to involve dysfunction of humoral and cellular immunity, producing a “cross-response” mediated by molecular mimicry between components of these pathogens and myelin or axons [4,5]. Its diagnosis is essentially clinical, although it is often complex due to its varied clinical presentation and evolution. However, lumbar puncture and electrodiagnostic testing can be useful for confirming or ruling out differential diagnoses [1,2].
Associated with motor and/or sensory symptoms that may result from peripheral nerve damage, signs of autonomic dysfunction are frequently observed, including fluctuations in blood pressure, cardiac arrhythmias, urinary retention, and respiratory failure [6].
Approximately 25% of patients require ventilatory support due to respiratory muscle compromise [7]. Although the mortality rate for Guillain-Barré syndrome is low (between 5 and 10% of cases) and the outcomes are generally favorable, 20% of patients have severe permanent disability, with gait disturbance and/or the need for ventilatory assistance [7,8]. This has a major impact on various aspects of the individual’s functioning, both in performing basic activities of daily living and in participating in work and social activities, resulting in a significant decline in quality of life [7]. As a result, the approach to these patients should not only focus on acute care and improving survival, but also on rehabilitation and reintegration into daily activities [7,9].
This article aims to provide a comprehensive and up-to-date summary of the main aspects to be addressed in the rehabilitation of patients with Guillain-Barré syndrome, offering general guidance for a multidisciplinary approach to these cases. To this end, a general summary of the clinical picture is presented. Next, the most relevant areas for a comprehensive rehabilitation process are reviewed. Finally, the main prognostic tools for this pathology are presented.
Methods
A search was conducted in databases such as MEDLINE/PubMed, Scopus, and PEDro Database, using key terms such as “Guillain-Barré syndrome,” “diagnosis,” “treatment,” “rehabilitation,” and “physical medicine.” The inclusion criteria were studies published in English or Spanish between 2015 and 2025 that addressed aspects of the comprehensive approach to Guillain-Barré syndrome. Articles published before 2015 were included only if they were pioneering studies on relevant aspects of rehabilitation management, for example, creating and validating a functional prognosis score.
Clinical features
Clinical evolution
Guillain-Barré syndrome typically presents with sensory impairment and progressive bilateral lower-limb motor weakness, which may extend to the upper limbs or trunk, accompanied by absent or diminished deep tendon reflexes in the affected limbs. The intensity of symptoms peaks between 2 and 4 weeks, followed by a plateau phase that can last from days to months. After this period, the patient begins to recover progressively, although recovery is not always complete [1,2] (Figure 1). Therefore, a subgroup of patients affected by Guillain-Barré syndrome will be left with varying degrees of long-term disability and sequelae of different kinds (weakness, fatigue, pain, dysautonomia, among others) [1,2,9].
Temporal course of Guillain-Barré syndrome.
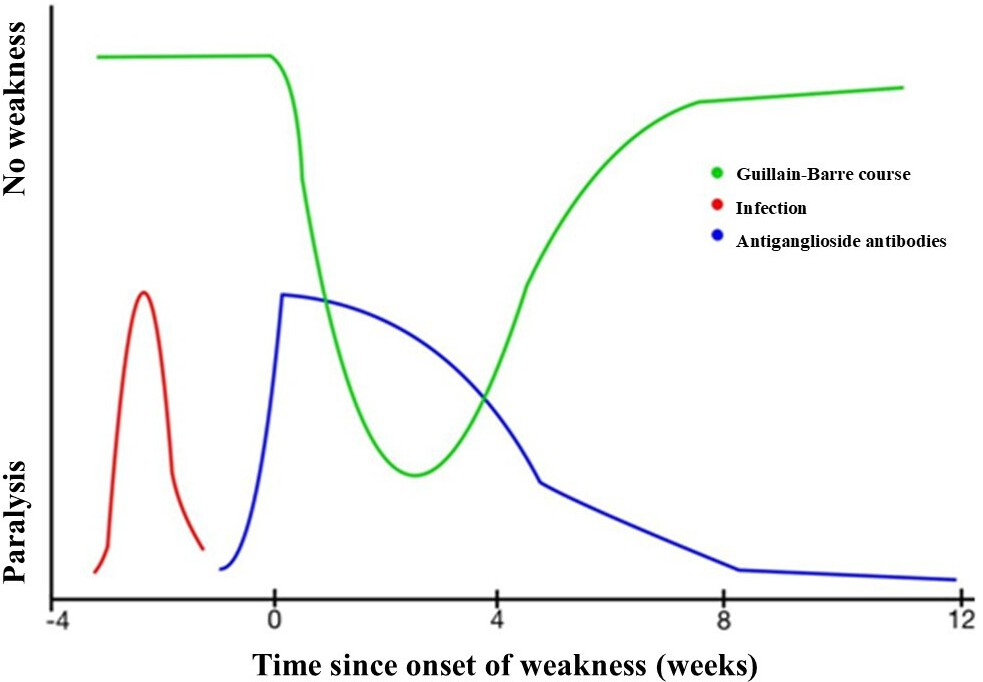
Guillain-Barré syndrome can exhibit different patterns, which are classified based on a combination of factors, including the topographical distribution of affected areas and the underlying pathophysiological alterations, detected by electrodiagnostic studies [1,2]. Among these described patterns, acute inflammatory demyelinating polyradiculoneuropathy (AIDP) is the most commonly reported form in published studies and has well-characterized clinical features and timing [10]. On the other hand, there is a variant of acute axonal motor neuropathy with pure motor axonal involvement and, at least clinically, the absence of detectable sensory alterations, as well as acute axonal motor and sensory neuropathy. Other variants that can be observed with Miller Fisher syndrome, whose classic triad includes ophthalmoparesis, ataxia, and areflexia, or the pharyngo-cervicobrachial variant, characterized by weakness of the pharyngeal, cervical, and upper limb muscles. Less common variants include Bickerstaff encephalitis, paraparesis, facial diplegia without limb weakness, or exclusive pandisautonomia [10,11] (Figure 2).
Clinical subtypes of Guillain-Barré syndrome.
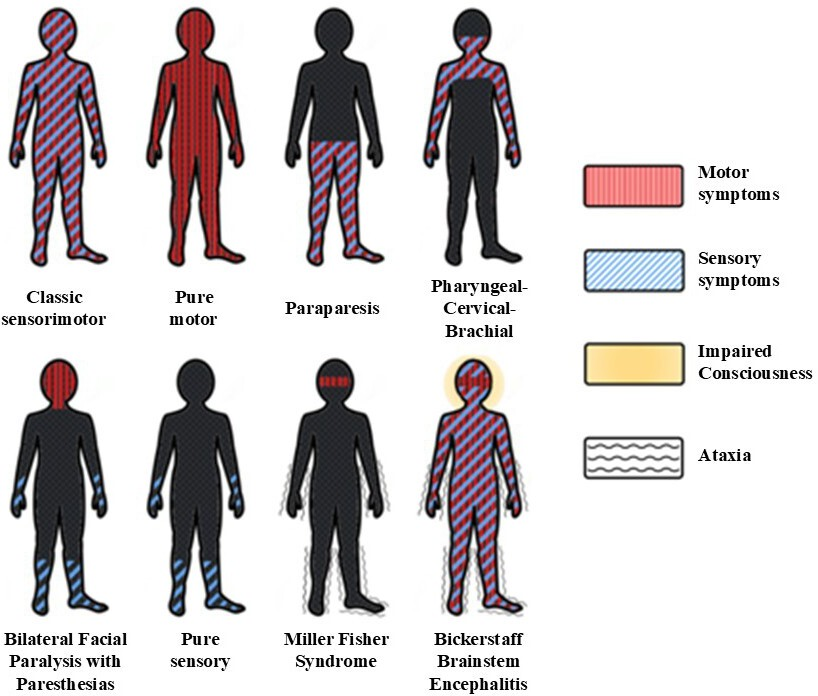
One-third of patients require admission to the intensive care unit, and this is associated with a significant risk of morbidity, mortality, and incomplete recovery. The main causes for admission to the intensive care unit are respiratory weakness and severe autonomic disturbances such as extreme bradycardia or cardiorespiratory arrest [12]. Other common symptoms during the course of Guillain-Barré syndrome are paresthesias (50%), cranial nerve involvement (up to 60% of patients), diarrhea/constipation (15%), syndrome of inappropriate antidiuretic hormone secretion (5%), and urinary retention (5%) [13,14].
Several conditions can mimic Guillain–Barré syndrome and therefore represent key differential diagnoses. In transverse myelitis, the presence of a sensory level and sphincter dysfunction, particularly urinary involvement, may help distinguish it. Myasthenia gravis can also present with weakness and cranial nerve involvement; however, it is typically characterized by fatigability as a cardinal feature, often accompanied by ptosis and ophthalmoplegia, with preserved deep tendon reflexes and fluctuating symptom severity. Additional diagnoses to consider include botulism, toxic–metabolic neuropathies, acute myopathies, chronic inflammatory demyelinating polyneuropathy, and multiple mononeuropathy [1,2,15]. A detailed discussion of the differential diagnostic approach to each of these entities is beyond the scope of this review.
The diagnosis of Guillain-Barré syndrome is based on clinical presentation, supported by complementary tests and procedures, such as lumbar puncture and electrodiagnosis, the detailed review of which is beyond the scope of this review. Standard treatment includes various supportive measures, depending on the observed dysfunctions and the severity of symptoms; for example, invasive mechanical ventilation in cases of respiratory failure (usually requiring a tracheostomy due to prolonged assisted ventilation) or an external pacemaker in cases of extreme bradycardia [12,16]. Specific treatments, such as intravenous immunoglobulin infusion or plasmapheresis, have led to reductions in recovery time and mortality in recent decades, although, as noted, a significant proportion of patients report residual disability that must be addressed comprehensively [16,17].
Disability and health-related quality of life in Guillain-Barré syndrome
Evidence published in recent years indicates that the impact of Guillain-Barré syndrome goes beyond acute motor impairment, significantly affecting patients' mental health and quality of life, even in the long term. Recent studies have shown that greater symptom severity, longer hospitalization, persistent pain, and advanced age are associated with poorer health-related quality-of-life outcomes, as assessed by instruments such as the Individualized Neuromuscular Quality of Life Questionnaire (INQoL), the EuroQoL-5D-5L, and the SF-36 [18,19,20].
Likewise, a high prevalence of anxiety, depression, and post-traumatic stress symptoms has been described in patients with Guillain-Barré syndrome, reinforcing the close relationship between the severity of the clinical course and mental health outcomes [18]. These factors not only influence patients' subjective perceptions of the disease but also have practical implications for hospital discharge planning and subsequent patient destinations, including the likelihood of discharge home or the need for more prolonged continuity of care in other institutions (e.g., long-stay centers) [21].
In this context, the medium- and long-term treatment of Guillain-Barré syndrome requires the participation of a multidisciplinary rehabilitation team that takes an integrated approach to motor and sensory deficits, autonomic symptoms, pain, and emotional and psychosocial aspects. Evidence suggests that a person-centered approach, particularly relevant in older patients, optimizes functional outcomes, community reintegration, and quality of life, especially when supported by integrative models such as the International Classification of Functioning, Disability, and Health (ICF), which will be presented later [18,19,20,21,22].
Comprehensive rehabilitation approach
Muscle weakness is usually a predominant finding in Guillain-Barré syndrome. Its extent and severity can range from total paralysis requiring mechanical ventilation to mild or moderate paresis of the dorsal foot flexors or the intrinsic hand muscles. Therefore, during motor rehabilitation, exercises may be passive, active-assisted, or active (with or without resistance) [16]. Occasionally, support may be required using devices such as anti-equinus orthosis (Figure 3) or resting pads to maintain proper joint resting position, so that, when muscle strength is recovered, functional use of these joints is possible [23,24,25,26]. In addition, during gait rehabilitation, along with technical aids such as walkers or canes, orthoses may be required to improve the function of certain joints whose active movement has been affected by paresis. Some of the alternatives used in cases of distal lower-limb paresis include ankle-foot orthoses, anti-steppage straps or foot-up orthoses, and ankle stabilizers (Figure 4) [27,28]. The use of these orthoses has been shown to improve walking speed and balance and to reduce energy expenditure, both in Guillain-Barré syndrome and in other motor-affecting polyradiculoneuropathies [27,28,29]. Prescription and orthotic fabrication should be performed after multidisciplinary evaluation by the rehabilitation team, considering potential functional benefits and other aspects, such as the actual feasibility of use, the risk of skin lesions, and cosmesis.
Anti-equinus orthosis.

Ankle-foot orthosis.
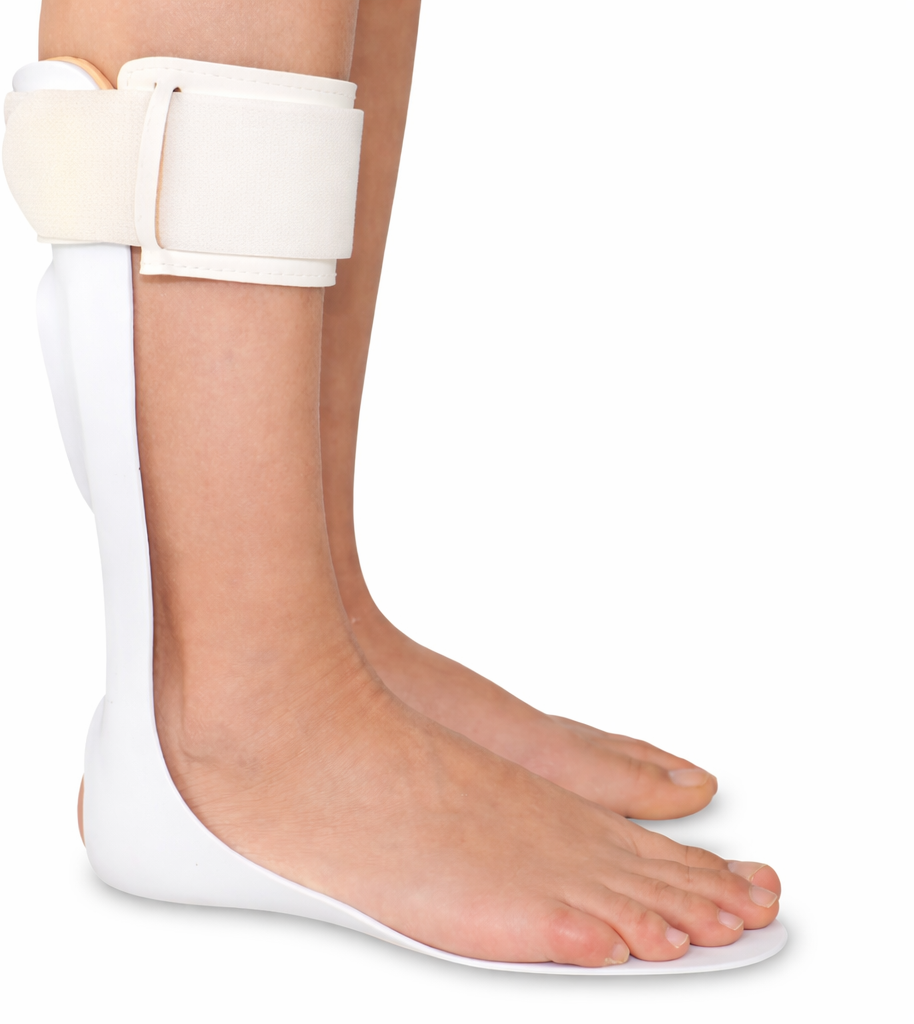
On the other hand, aerobic training using equipment such as upper-limb cycle ergometers or bicycles has been shown to reduce fatigue and improve physical condition and quality of life in patients with Guillain-Barré syndrome. The above exercises should be combined with strength and balance exercises, as well as postural control training [16,30,31]. In addition, benefits have been reported in the motor rehabilitation of patients with Guillain-Barré syndrome with the use of hydrotherapy and functional electrical stimulation [32,33].
Another alteration that can be observed in these patients is weakness of the respiratory muscles. In this regard, respiratory muscle strength can be assessed using measurements such as maximum inspiratory and maximum expiratory pressures [34,35]. These values can be used to determine whether the patient has an abnormality compared to a normal population, as well as to plan respiratory muscle training, in which threshold valves can be used. The benefit of these interventions has been proposed in a small feasibility study on Guillain-Barré syndrome and in a meta-analysis, based on observations in various neuromuscular diseases [34,35]. Another assessment used in exercise programs for patients with Guillain-Barré syndrome is grip strength measured with a dynamometer, either during follow-up or as an independent predictor of overall functional status [36]. The participation of the nutrition team in nutritional assessment and the adjustment of intake according to patient requirements is essential, especially during the acute hospital period. This intervention can impact the functional outcome of patients with Guillain-Barré syndrome [37]. In this regard, a retrospective study showed that poorer nutritional status in patients with Guillain-Barré syndrome is associated with greater disability, reduced functional independence, and longer stays in rehabilitation. Likewise, improvements in nutritional status during rehabilitation were associated with more favorable functional recovery, underscoring the importance of nutritional assessment as part of a comprehensive approach to Guillain-Barré syndrome [37].
Finally, an important aspect to consider during the rehabilitation process is the training of family members and caregivers [26]. The aim is, on the one hand, to educate them on proper methods for assisting patients during transfers and transitions to reduce the risk of injury and musculoskeletal pain among caregivers, and, on the other hand, to reduce the incidence of such injuries and pain among caregivers.
Autonomic dysfunction and pain
Autonomic dysfunction is caused by small fiber compromise. Symptoms include decreased sweating (which could lead to skin alterations and, together with sensory deficit, cause pressure injuries), orthostatic hypotension, gastroparesis or constipation, urinary retention, among others [6,14]. As for orthostatic hypotension, bipedal tolerance can be improved by training on a tilt table [38] (Figure 5), combined with abdominal belts or graduated compression stockings on the lower extremities to promote venous return and consequently improve bipedal tolerance. In addition, there is evidence, although not conclusive, of the use of functional electrical stimulation for this purpose [39].
Tilting table for orthostatic training.
Pharmacological and non-pharmacological interventions may be required for pain management. Regarding the former, consideration should be given to the presence of musculoskeletal pain resulting from muscle imbalance and secondary myofascial syndrome, shortening and contractures, together with the exacerbation of previous pathologies such as tendinopathies or osteoarthritis. It is also necessary to observe the onset of neuropathic pain in more than 50% of patients with Guillain-Barré syndrome [16,40,41]. Consequently, the use of neuromodulatory drugs as adjuncts to standard analgesic drugs is common. The most commonly used drug groups include dual antidepressants, gabapentinoids, and anticonvulsants [40,41]. For non-pharmacological management, interventions such as desensitization therapies, motor imagery, mindfulness, and transcutaneous electrical stimulation can be employed. However, their benefits are extrapolated from studies of other types of neuropathic or chronic pain [42,43].
Neurogenic bladder and bowel
Bladder dysfunction occurs in several neurological disorders. In Guillain-Barré syndrome, it can be present in up to 30% of cases, and its presentation varies from incontinence to incomplete emptying or bladder distension, among other manifestations [6,44]. The most serious complications include upper urinary tract infection with secondary kidney damage and the appearance of skin lesions secondary to excessive moisture and skin maceration in cases of incontinence.
When cases of neurogenic bladder occur in Guillain-Barré syndrome, up to 10% are expressed as urinary retention [44]. Because of this, many patients may require the use of a urinary catheter during the first few days to prevent bladder overdistension and vesicoureteral reflux. In addition, this device measures diuresis and calculates water balance. However, when the patient is stable and in cases where bladder dysfunction persists, it is advisable to initiate intermittent urinary catheterization, as this allows for a bladder distension/evacuation rhythm closer to the physiological pattern and has also been shown to result in a lower rate of infectious complications during follow-up. In cases of persistent dysfunction, a formal urodynamic study is useful [45].
In addition, some patients with Guillain-Barré syndrome may experience paresis or dysfunction of the pelvic floor muscles and may therefore benefit from targeted training therapies [46]. This intervention can be combined with techniques such as electrical stimulation or biofeedback. In rare cases of overactive bladder, alternatives include anticholinergic agents or botulinum toxin injection into the detrusor muscle [45,46].
Among patients with Guillain-Barré syndrome who present with neurogenic bowel, the most common disorder is constipation, although cases of diarrhea and even adynamic ileus have been reported [14,47]. Its approach includes non-pharmacological measures such as adequate hydration, a high-fiber diet, and maintenance of regular bowel movement schedules. However, some patients may require the use of drugs such as lactulose or polyethylene glycol to achieve regular, painless bowel movements. Although these interventions have not been specifically studied in Guillain-Barré syndrome, they are often extrapolated from the management of patients with neurogenic bowel of other origins, such as spinal cord injury [48].
When treating neurogenic bladder and bowel disorders, it is always necessary to consider the impact these disorders can have on patients' quality of life. In follow-up studies lasting more than six years, 49% of patients with Guillain-Barré syndrome who continued to experience urinary symptoms reported interference with their daily lives, and 10.6% reported a significant impact on their quality of life [49]. It has been observed that the decline in self-esteem and the embarrassment generated by procedures such as intermittent urinary catheterization can have a significant impact on family, social, and work reintegration. Therefore, therapeutic support from the rehabilitation team, including psychological support, is essential for this group of patients.
Residual fatigue in Guillain-Barré syndrome
Although many patients show favorable recovery, particularly in muscle strength, a recent meta-analysis reported that 22% to 44% of patients experience fatigue during follow-up, which can adversely affect their quality of life [50]. In fact, fatigue is one of the three most disabling symptoms, according to patients themselves. A prospective study found that fatigue was significantly more frequent and severe among patients aged over 50 years and women, with no statistically significant association with a clinical history of viral or bacterial infection. No association was found between serologic evidence of infection and infection in patients in whom it was identified [51].
Although the causes of fatigue are not fully understood, central alterations, peripheral nerve physiology dysfunction, and cognitive-emotional aspects have been proposed [52,53]. The management of fatigue includes educating patients on energy conservation and joint protection techniques, promoting lifestyle changes (e.g., rhythm regulation, planned breaks), and improving sleep hygiene. Likewise, in Guillain-Barré syndrome and other neurological disorders, modifications to the home environment may be considered to enhance safety and accessibility. Examples include non-slip surfaces, grab bars or handrails, lighting adjustments, and clearing hallways, among others [54]. On the other hand, in cases of severe fatigue, even without a priori severe balance disorder, the combination of indoor walking aids (e.g., a walker or canes) with a wheelchair for community mobility should be considered, as the walking pattern may worsen as fatigue progresses. Finally, fatigue is a symptom that must be taken into account when planning aspects such as returning to driving and reintegration into work and society.
Technical assistance
The selection of a walking aid depends, among other things, on the patient’s strength, stability, coordination, cardiovascular capacity, and cognitive status [55]. There are various standardized tests for stroke and other acquired brain injuries that assess balance and can aid decision-making [56]. The alternatives range from devices that provide maximum stability and support, such as a walker, to others that allow greater freedom but offer less relative stability, such as a cane. On the other hand, given that many patients continue to experience fatigue or have had their cardiopulmonary capacity affected as a result of Guillain-Barré syndrome or the decompensation of their comorbidities, some patients may need to use a wheelchair for long journeys and trips outside the home. This choice will take into account factors such as the patient’s trunk and pelvic control, upper-limb function, body weight, and the terrain on which the technical aid will be used most often, among others [55].
Rehabilitation plan
After considering the above aspects, a rehabilitation plan is established based on an accurate assessment of current functional status, the severity of Guillain-Barré syndrome, and its prognosis. Following this comprehensive assessment, a list of factors affecting the subject’s functioning is usually compiled, summarized in the Trimodal Diagnosis of the International Classification of Functioning, Disability, and Health model [57] (Figure 6).
Conceptual framework of the International Classification of Functioning, Disability, and Health (ICF).
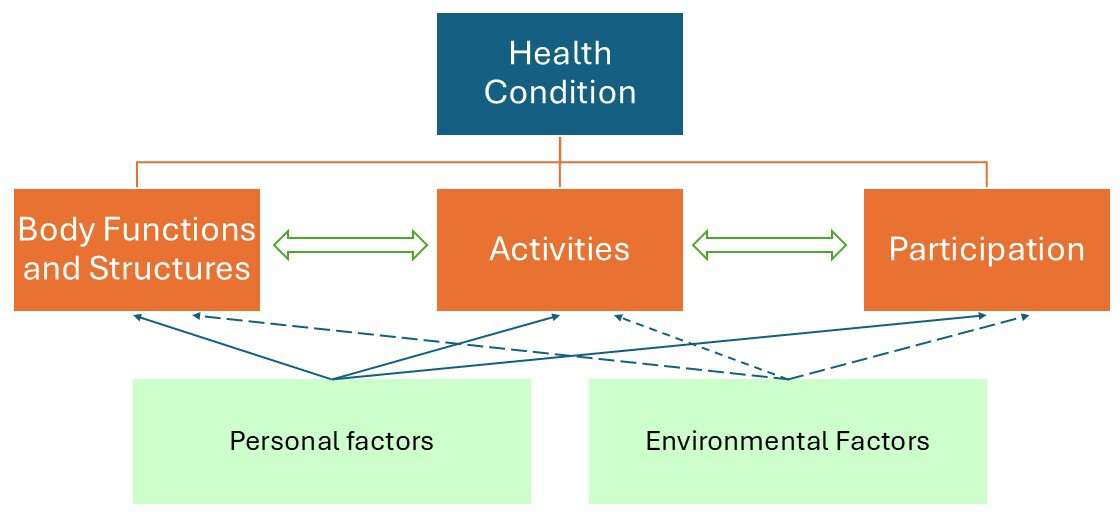
The rehabilitation plan addresses physical deficiencies (weakness, contractures, pain, etc.) to improve function and prevent progression. On the other hand, remaining abilities must be trained in a targeted manner with the same objective. When necessary, functional independence can be achieved with technical aids and environmental adaptations, as described above. Thus, depending on the functional objectives and the interventions proposed to achieve them, the different members of the rehabilitation team participate in an orderly and coordinated manner during this process [57,58,59]. The rehabilitation plan should be reviewed on an ongoing basis through regular multidisciplinary meetings to update the objectives and interventions. This model of case management can have a positive impact on functional recovery, compared to partial or unstructured approaches [59].
Follow-up
Considering the residual disability that affects many patients with Guillain-Barré syndrome at the time of discharge, it is essential to have medical follow-up systems and outpatient rehabilitation therapies in place to ensure continuity of care. In addition, complications such as the onset of pain, mood disorders, or caregiver overload often manifest with greater intensity during the outpatient rehabilitation period. For these reasons, structured follow-up programs are an opportune time for early detection and intervention [60].
Various instruments are used to establish the neurological prognosis in Guillain-Barré syndrome. Some focus on the likelihood of respiratory failure, while others focus on the progression of the disease and the prediction of functional recovery [61]. Functional outcome is commonly assessed using the Guillain-Barré syndrome disability scale, also known as the Hughes scale [61]. This scale assesses the functional status of patients with Guillain-Barré syndrome. It has seven levels, scored from 0 to 6, which describe increasing degrees of disability, from healthy patients to deceased patients. Levels 0 to 2 are generally considered to indicate an acceptable degree of functional independence, although patients at level 3 can walk with technical assistance [61]. Another useful and validated instrument is the Erasmus Guillain–Barré Syndrome Respiratory Insufficiency Score (EGRIS) (Table 1). It is used to estimate the risk of a patient developing respiratory failure and requiring mechanical ventilation within the first seven days of hospital admission [61,62]. Predictions are based on three clinical factors determined upon hospital admission: the time from onset of weakness to admission, the presence of facial and/or bulbar weakness, and the severity of muscle weakness, defined by the total score on the Medical Research Council sum score (MRC-ss) scale [61,62]. The total EGRIS score ranges from 0 to 7, corresponding to an estimated risk of respiratory failure within the first week ranging from 1 to 90% [62].
Another important tool is the Erasmus GBS Outcome Score (EGOS), with its modified version (Table 2) [63]. This clinical prognostic model predicts patients' independent or modified walking ability in Guillain-Barré syndrome. In practice, it refers to any degree of walking, not necessarily long-distance walking within the community. The factors included in this scale are age, presence of diarrhea, and MRC-ss score. It can be performed upon hospital admission; however, if performed seven days after admission, it has greater predictive power. When the collected data are entered into the calculator, it yields the probability of not being able to walk independently at 1, 3, and 6 months of follow-up [63]. For example, an EGOS score of 4 points at hospital admission predicts that the patient will be unable to walk without assistance, with probabilities of 38% and 10% at one month and three months of follow-up, respectively. In other words, they have a 62% and 90% probability of achieving independent or modified independent walking at one month and three months, respectively.
The relevance of these prognostic tools lies in guiding decision-making using rapidly obtained clinical information. For example, a high EGRIS score could define early transfer to a critical care unit, given the high risk of requiring invasive mechanical ventilation. On the other hand, a low EGOS score enables planning of environmental interventions and the indication of technical aids, assuming a high probability of independent or modified independent walking in the medium term.
Conclusions
Guillain–Barré syndrome is a potentially disabling condition with a significant impact on the functioning and quality of life of patients who receive this diagnosis. Traditionally, motor symptoms have been the most widely recognized and addressed. However, throughout this article, in addition to the consequences of impaired muscle strength, other equally relevant aspects for the rehabilitation process have been discussed, including pain, autonomic symptoms, and emotional and psychosocial factors.
Rehabilitation planning for these patients is highly challenging and requires a coordinated, multidisciplinary approach in which the actions of the professionals involved converge while actively incorporating the patient and their environment. A useful framework for a comprehensive assessment of the factors contributing to disability in patients with Guillain–Barré syndrome is the International Classification of Functioning, Disability and Health (ICF) model, as it considers not only the health condition (medical diagnosis) but also the evaluation of impairments, activity limitations, and participation restrictions, together with personal and contextual factors, whose relevance cannot be overlooked.
Current evidence shows that disability may persist for years after the onset of this condition, making it necessary to design medium- and long-term clinical follow-up strategies. Likewise, once the acute phase has resolved and functional independence improves, additional challenges arise in the rehabilitation process, including return to work and community reintegration.
Finally, the existence of a knowledge gap should be acknowledged: there are few high-quality methodological studies on the most effective rehabilitation strategies for Guillain–Barré syndrome, and clinical practice guidelines specifically addressing this topic are absent. Nevertheless, this article synthesizes the key considerations in the rehabilitation process, providing general guidance for the comprehensive management of these patients.

