Últimos artículos publicados
Control metabólico de pacientes diabéticos tipo 2 tratados con empagliflozina
Wilfredo Navarrete, et al. (17/04/2024)
Revisión de literatura sobre sexualidad en las personas mayores: qué se educa y con qué tecnologías
Jhonny Acevedo Ayala, et al. (15/04/2024)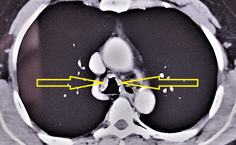
Traqueobroncopatía osteocondroplástica
Luis Alejandro Rodríguez Hidalgo, et al. (08/04/2024)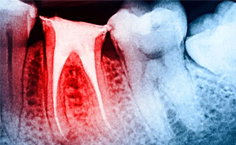
Características clínicas y radiográficas de reabsorciones radiculares externas
Agustina Buchi-Velazquez, et al. (04/04/2024)
Sistemas de acreditación de programas de formación en partería profesional en el mundo
Jovita de las Rozas Ortiz-Contreras, et al. (02/04/2024)
Telesalud mental en una unidad pública de psiquiatría infanto-juvenil en pandemia
Francisca García, et al. (21/03/2024)Información
Colecciones
Artículos más leídos
Trastornos del ritmo cardíaco: electrofisiología, arritmias benignas
Claudia García, et al. (01/08/2009)