Resúmenes Epistemonikos
← vista completaPublicado el 11 de octubre de 2018 | http://doi.org/10.5867/medwave.2018.06.7286
Cannabinoides para el tratamiento del trastorno por abuso de cannabis
Cannabinoids for the treatment of cannabis abuse disorder
Resumen
INTRODUCCIÓN El cannabis se erige como la droga ilícita más consumida en el mundo. Actualmente no existen alternativas farmacológicas específicas para el tratamiento de su adicción, por lo que se ha postulado la utilidad del uso de cannabinoides como herramienta terapéutica. Ellos actuarían principalmente a través de la disminución de síntomas de abstinencia y craving (deseo o compulsión por consumir), pero su efectividad aún no está clara.
MÉTODOS Para responder esta pregunta utilizamos Epistemonikos, la mayor base de datos de revisiones sistemáticas en salud a nivel mundial, la cual es mantenida mediante búsquedas en múltiples fuentes de información, incluyendo MEDLINE, EMBASE, Cochrane, entre otras. Extrajimos los datos desde las revisiones identificadas, reanalizamos los datos de los estudios primarios, realizamos un metanálisis y preparamos una tabla de resumen de los resultados utilizando el método GRADE.
RESULTADOS Y CONCLUSIONES Identificamos siete revisiones sistemáticas que en conjunto incluyeron 15 estudios primarios, de los cuales cuatro corresponden a ensayos aleatorizados. Concluimos que el uso de cannabinoides podría resultar en poco o nulo aumento en la abstinencia al finalizar el tratamiento, y probablemente aumenta los efectos adversos.
Problema
El trastorno por abuso de sustancias es un problema epidemiológicamente relevante que se define por el desarrollo de patrones conductuales desadaptativos en relación al consumo de alguna sustancia, y que en general se acompaña de tolerancia, uno de los elementos diagnósticos de la dependencia. En este contexto, el cannabis se erige como una de las drogas ilícitas con potencial adictivo más consumidas en el mundo [1].
Si bien no existen alternativas farmacológicas específicas para el tratamiento del trastorno por abuso de cannabis, diversos estudios han postulado que el sistema endocannabinoide tiene un rol en la modulación de varios circuitos neuronales asociados a la adicción a drogas, y en este contexto se ha propuesto la utilización de cannabinoides como alternativa terapéutica en pacientes que sufren de trastorno por consumo de cannabis. Al igual que la terapia de reemplazo de nicotina se utiliza comúnmente para el cese del hábito tabáquico, se postula el uso de cannabinoides para la disminución de los síntomas de abstinencia y craving.
Métodos
Para responder esta pregunta utilizamos Epistemonikos, la mayor base de datos de revisiones sistemáticas en salud, la cual es mantenida mediante búsquedas en múltiples fuentes de información, incluyendo MEDLINE, EMBASE, Cochrane, entre otras. Extrajimos los datos desde las revisiones identificadas y reanalizamos los datos de los estudios primarios. Con esta información, generamos un resumen estructurado denominado FRISBEE (Friendly Summaries of Body of Evidence using Epistemonikos), siguiendo un formato preestablecido, que incluye mensajes clave, un resumen del conjunto de evidencia (presentado como matriz de evidencia en Epistemonikos), metanálisis del total de los estudios cuando sea posible, una tabla de resumen de resultados con el método GRADE y una sección de otras consideraciones para la toma de decisión.
|
Mensajes clave
|
Acerca del conjunto de evidencia para esta pregunta
|
Cuál es la evidencia |
Encontramos siete revisiones sistemáticas [2],[3],[4], |
|
Qué tipo de pacientes incluyeron los estudios* |
Tres ensayos [9],[11],[12] incluyeron pacientes adultos con diagnóstico de dependencia a cannabis según criterios DSM - IV- TR, y uno [10] incluyó pacientes descritos como dependientes de cannabis reclutados de la comunidad sin especificar el criterio de dependencia. Tres ensayos [9],[11],[12] excluyeron pacientes que tuvieran comorbilidades psiquiátricas significativas o dependencia de otras sustancias (exceptuando nicotina y cafeína), y en uno no se especificó criterios de exclusión de los pacientes. |
|
Qué tipo de intervenciones incluyeron los estudios* |
Dos ensayos evaluaron nabiximol (Sativex) como intervención durante 6 días [9] y durante 8 semanas [10]. Un ensayo [11] utilizó dronabinol vía oral como monoterapia y otro [12] utilizo dronabinol asociado a lofexidina (un agonista alfa-2 adrenérgico). Además, un ensayo [9] utilizó en paralelo terapia cognitivo conductual en ambos grupos. |
|
Qué tipo de desenlaces midieron |
Los ensayos evaluaron múltiples desenlaces, los cuales fueron agrupados por las distintas revisiones sistemáticas de la siguiente manera:
|
* La información sobre los estudios primarios es extraída desde las revisiones sistemáticas identificadas, no directamente desde los estudios, a menos que se especifique lo contrario.
Cuál es la evidencia
Resumen de los resultados
La información sobre los efectos de cannabinoides está basada en cuatro ensayos aleatorizados que incluyen 338 pacientes [9],[10],[11],[12].
Un ensayo midió el desenlace abstinencia al fin del tratamiento (156 pacientes) [11], cuatro ensayos midieron el desenlace síntomas de abstinencia y craving (338 pacientes) [9],[10],[11],[12] y un ensayo midió el desenlace efectos adversos (156 pacientes) [11]. Respecto a los síntomas de abstinencia/craving, ninguna revisión permitió la extracción de datos de manera que pudieran ser incorporados a un metanálisis, por lo que la información de dicho desenlace se presenta como síntesis narrativa.
El resumen de los resultados es el siguiente:
- El uso de cannabinoides podría resultar en poco o nulo aumento en la abstinencia al fin del tratamiento, pero la certeza de la evidencia es baja.
- No está claro si el uso de cannabinoides disminuye los síntomas de abstinencia y craving porque la certeza de la evidencia es muy baja.
- El uso de cannabinoides probablemente aumenta los efectos adversos. La certeza de la evidencia es moderada.
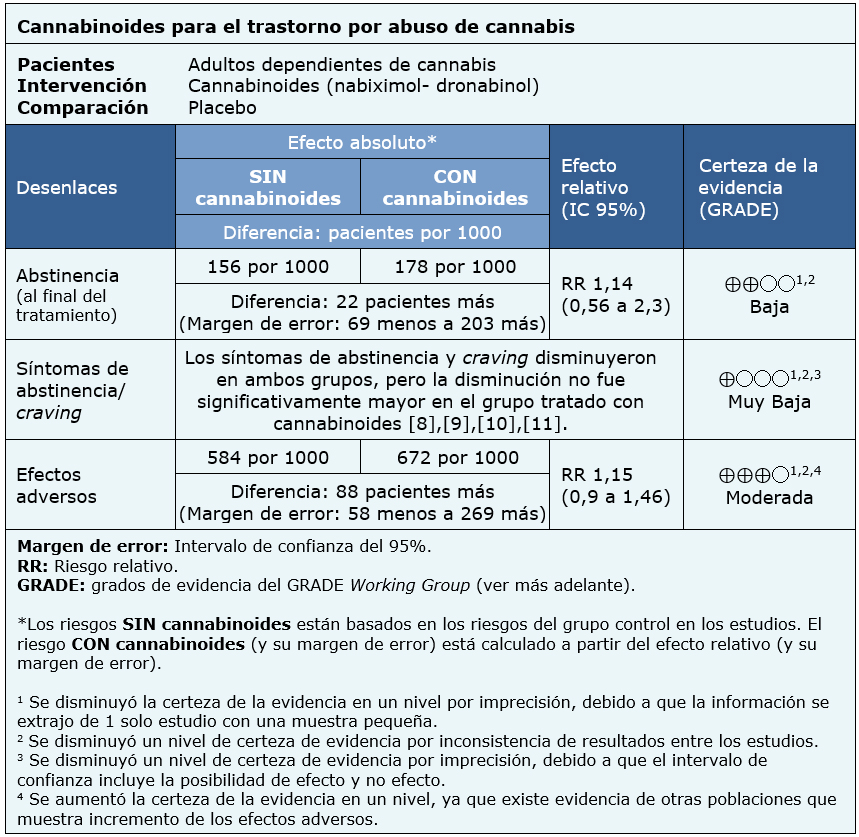
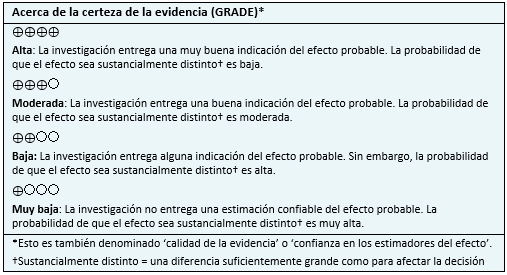
| Siga el enlace para acceder a la versión interactiva de esta tabla (Interactive Summary of Findings - iSoF) |
Otras consideraciones para la toma de decisión
|
A quién se aplica y a quién no se aplica esta evidencia |
|
| Sobre los desenlaces incluidos en este resumen |
|
| Balance riesgo/beneficio y certeza de la evidencia |
|
| Consideraciones de recursos |
|
| Qué piensan los pacientes y sus tratantes |
|
| Diferencias entre este resumen y otras fuentes |
|
| ¿Puede que cambie esta información en el futuro? |
|
Cómo realizamos este resumen
Mediante métodos automatizados y colaborativos recopilamos toda la evidencia relevante para la pregunta de interés y la presentamos en una matriz de evidencia.
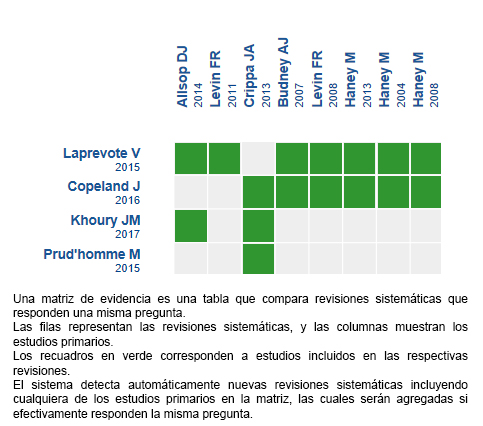
Siga el enlace para acceder a la versión interactiva: Cannabinoides para el trastorno por consumo de cannabis.
Notas
Si con posterioridad a la publicación de este resumen se publican nuevas revisiones sistemáticas sobre este tema, en la parte superior de la matriz se mostrará un aviso de “nueva evidencia”. Si bien el proyecto contempla la actualización periódica de estos resúmenes, los usuarios están invitados a comentar en la página web de Medwave o contactar a los autores mediante correo electrónico si creen que hay evidencia que motive una actualización más precoz.
Luego de crear una cuenta en Epistemonikos, al guardar las matrices recibirá notificaciones automáticas cada vez que exista nueva evidencia que potencialmente responda a esta pregunta.
Este artículo es parte del proyecto síntesis de evidencia de Epistemonikos. Se elabora con una metodología preestablecida, siguiendo rigurosos estándares metodológicos y proceso de revisión por pares interno. Cada uno de estos artículos corresponde a un resumen, denominado FRISBEE (Friendly Summary of Body of Evidence using Epistemonikos), cuyo principal objetivo es sintetizar el conjunto de evidencia de una pregunta específica, en un formato amigable a los profesionales clínicos. Sus principales recursos se basan en la matriz de evidencia de Epistemonikos y análisis de resultados usando metodología GRADE. Mayores detalles de los métodos para elaborar este FRISBEE están descritos aquí (http://dx.doi.org/10.5867/medwave.2014.06.5997)
La Fundación Epistemonikos es una organización que busca acercar la información a quienes toman decisiones en salud, mediante el uso de tecnologías. Su principal desarrollo es la base de datos Epistemonikos (www.epistemonikos.org).
Declaración de conflictos de intereses
Los autores declaran no tener conflictos de intereses con la materia de este artículo.

